
Differential Interference Contrast (DIC) Microscopy
and other methods of producing contrast
by Dr. Robert Berdan
May 12, 2021
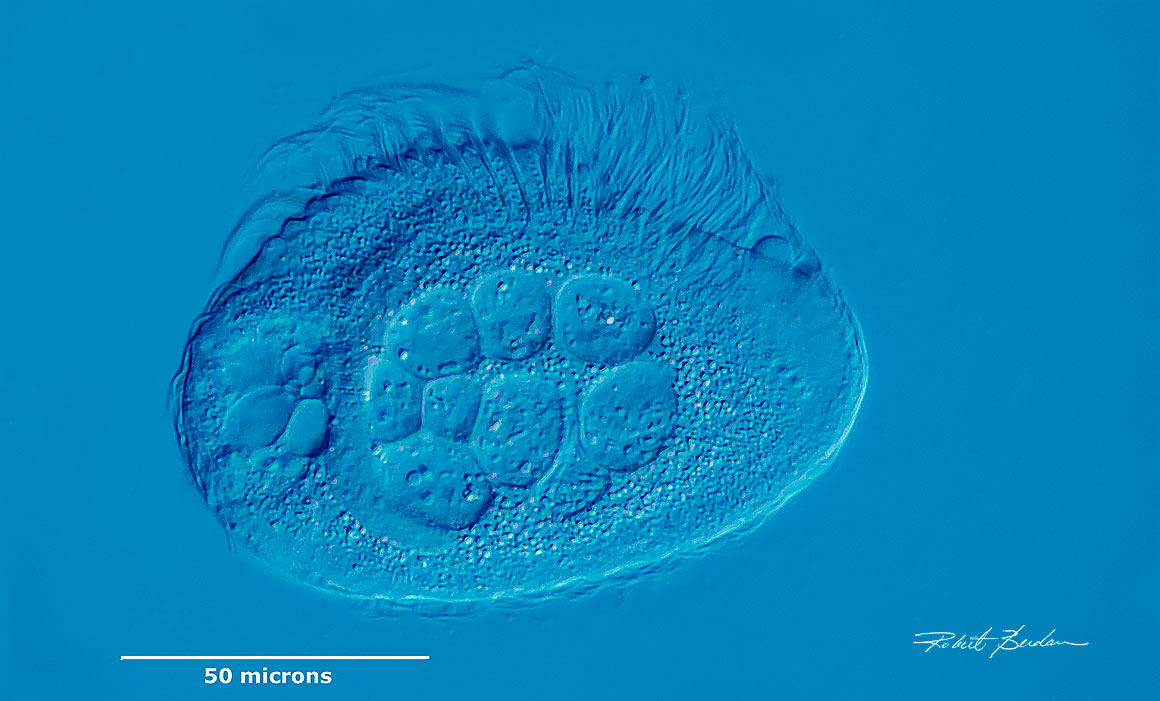
Phacodinium metchnikoffi is a ciliate found within water in moss. Photographed by Differential Interference Contrast (DIC) microscopy. Note the pseudo 3D relief and fine detail.
Introduction
One of the challenges microscopists face when studying live cells is the lack of contrast as cells are made up of about 70% water. Scientists first stained cells and bacteria with water soluble dyes to make them more visible in the microscope. Unfortunately many dyes are toxic to the cells, or require that the cells be fixed in alcohol and mounted on a glass slide. This eliminates the possibility to study dynamic activities such as cell behaviour and cell division. Microscopy techniques that are employed to provide contrast include: dark-field, phase contrast, polarization, fluorescence, differential interference contrast (DIC), Hoffman modulation contrast, and oblique lighting. Sometimes several techniques are combined. I show pictures using each technique, discuss some of their pros and cons and describe how DIC microscopy works.
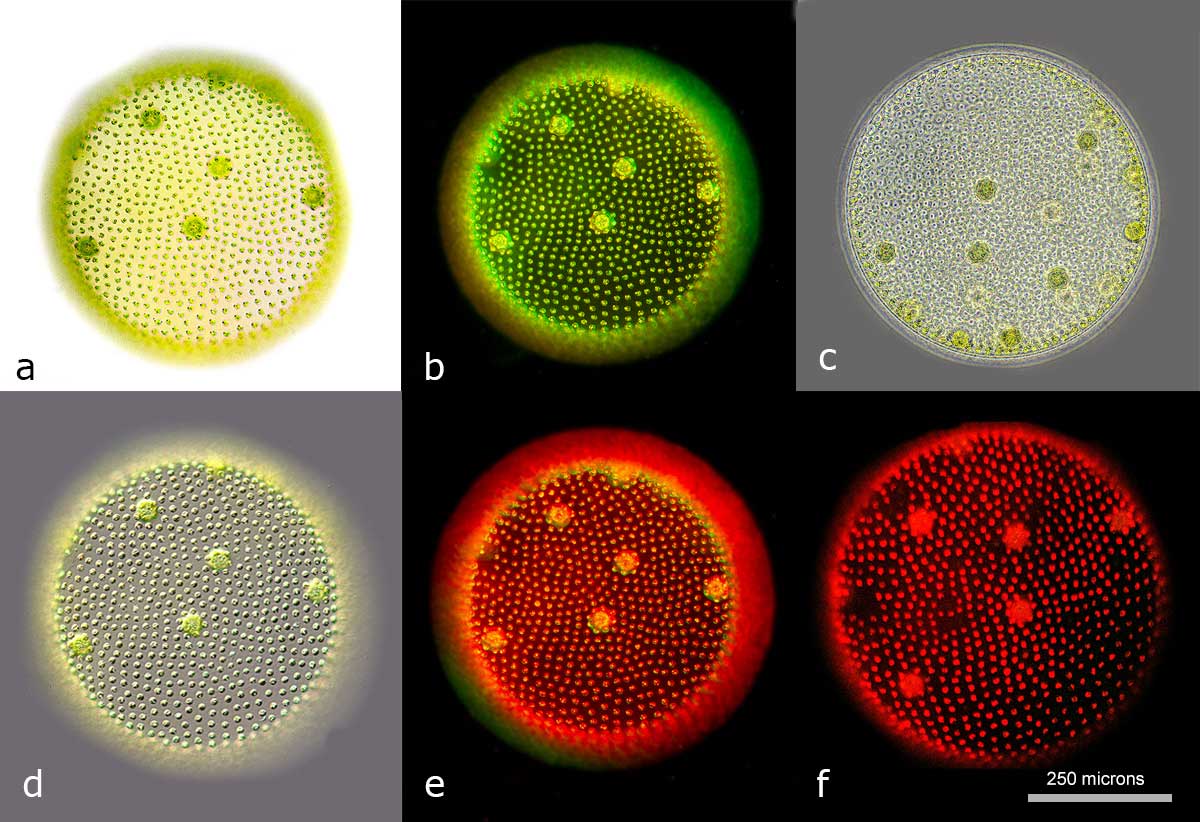
Volvox, a green algae living in fresh water ponds viewed by different kinds of microscope illumination a) bright-field b) dark-field c) phase contrast d) differential Interference contrast e) Rheinberg lighting f) fluorescence microscopy with green excitation.
Table of Contents
1. Bright-field microscopy
2. Dark-field microscopy
3. Rheinberg contrast
4. Phase contrast microscopy
5. Polarized light microscopy
6. Fluorescence light microscopy
7. Differential Interference microscopy
8. Hoffman modulation contrast microscopy
9. Oblique Lighting microscopy
10. Simulate DIC and Phase contrast microscopy with software
11. Summary
12. References
Bright-field microscopy
Bright-field microscopy is the standard form of illumination that most light microscopes offer and the least expensive mode. Bright-field is great for studying stained histological slides, ciliates, bacteria and parasites. Some live microorganisms are coloured by pigments or contain symbiotic organisms (e.g. algae) as shown in the picture below. Most living cells are almost transparent and low in contrast. Lowering the microscope condenser under the stage or reducing the condenser diaphragm can improve contrast and improve intracellular details but it also lowers resolution. Bright-field microscopy also works well with living specimens that can be stained with vital dyes or arthropods that have a thin transparent cuticle made of chitin.

Stentor polymorphus a single cell ciliate found in pond water with intracellular symbiotic algae (Chlorella sp). The Macro nucleus, external cilia, and other intracellular organelles are sometimes difficult to see with bright field microscope lighting.

Above are live human cheek epithelial cells stained with Methylene Blue. The large dark blue oval shaped structures are nuclei. The smaller pill shaped structures are bacteria on the cell surface. Cells are harvested by scraping the inside of the mouth with another glass slide and then spreading the them on a second glass slide and after staining and rinsing covering the cells are covered with a cover-slip. 400X Bright–field microscopy. Cheek cells are useful for testing because you always have some with you and they are easy to extract.
Dark-field Microscopy
Dark-field microscopy illuminates the specimen with a cone of light and the specimen refracts some of the light up into the objective. This technique can be applied using a dark-field condenser or metal stop (e.g. coin) that fits below the condenser in the light path. Some phase contrast and DIC condensers include a dark-field stop which works well for 10, 20X, 40X objectives but is less effective for 60X or 100X objectives. Darkfield condensers are also available by most microscope companies and start around $100 and up. Live cells, Cladocerans (e.g. Daphnnia), ciliates, and some bacteria exhibit good contrast by this technique. Fritz Schaudinn and Eric Hoffman discovered the bacteria Treponema pallidum that causes syphilis in 1905 using dark-field microscopy. I will present a separate article on dark-field microscopy in the future.
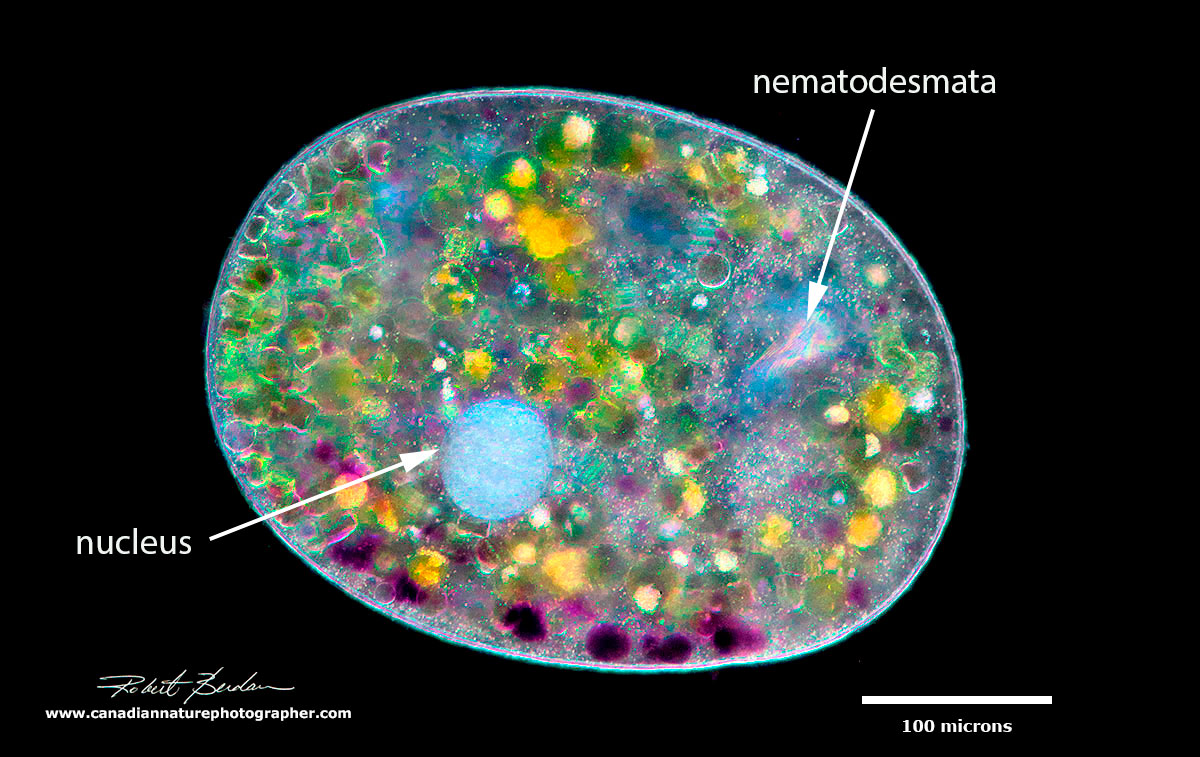
Above is a Ciliate - Nassophorean Nassula ornata - its mouth leads to palisade of rods called nematodesmata seen inside the cell at the upper right (bright white area) and this ciliate has a single macro-nucleus (white circle). This organism feeds on blue-green algae and as the algae are broken down they take on a variety of bright colours from green to purple. Dark-field microscopy 400X - panorama stitch.
Rhineberg filters to produce colour Contrast
Rheinberg filters behave in a manner similar to dark-field in that the outer colour of the filter illuminates the refracted light while the center colour of the filter lights the background. These filters can be purchased on Ebay.com for $25-40 a set or made at home using a laser printer or cutting coloured vinyl sheets (J. Rheinberg, 1896, Shaw 2011R , A.A. Havics, 2014, R. Berdan, 2017 ). Almost any microscope can be modified to work with these filters. I made a small card board box with a hole in it to position the filters the optimum height below the condenser of my microscope and they can also be hand-held.

Fresh water rotifer, Synchaeta sp viewed by Rheinberg Lighting combined with dark-field microscopy.
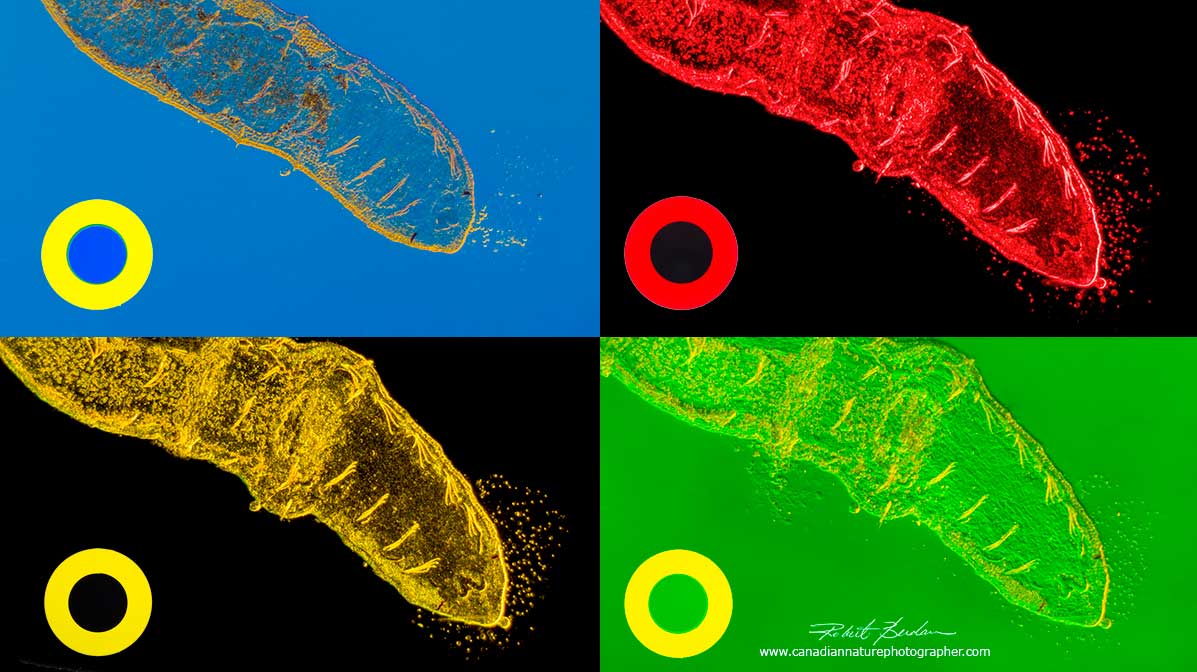
Aquatic ribbon worm photographed with different coloured Rheinberg filters. Each filter is shown in the lower left corner of each picture and made of an outer and inner color. 50X. Rheinberg filters work well from 50 to 400X.
Phase contrast
One of the best techniques to add contrast to live cells with a microscope is phase contrast invented by Fritz Zernike who received the Nobel Prize in Physics in 1963 for its invention. Phase contrast can be added to most microscopes for less than $1500 depending on the manufacturer and quality of the condenser and objectives. The two main elements needed are the objectives which have special rings inside them and the condenser which also has a special matching ring for each objective. The rings must be aligned and oriented with a phase telescope (R. Berdan 2021). A phase telescope is used to align the objective and condenser rings. There are two main types of phase contrast: positive and negative. Positive phase contrast is the most popular type and produces a light background and high refractive index organelles are dark in colour (see yeast cell below). Negative phase has a dark background and more refractile cell components appear bright (see below). The main disadvantage of phase contrast is that thick specimens can be difficult to interpret and it produces small halos around the cells components which slightly reduce the overall resolution. Phase contrast is one of the best techniques to study living cells and aquatic organisms with a light microscope. Anyone looking to upgrade their bright field microscope should consider adding phase contrast. To understand how phase contrasts works see R. Berdan (2021). For general purposes I recommend the positive phase contrast.
Yeast cells of Saccharomyces cerevisiae are used in baking and beer brewing. Phase contrast 1000X oil immersion. Note the outer white halo which is an artifact of the technique. This is image was photographed using positive phase contrast microscopy with a 50 year old Olympus E microscope.
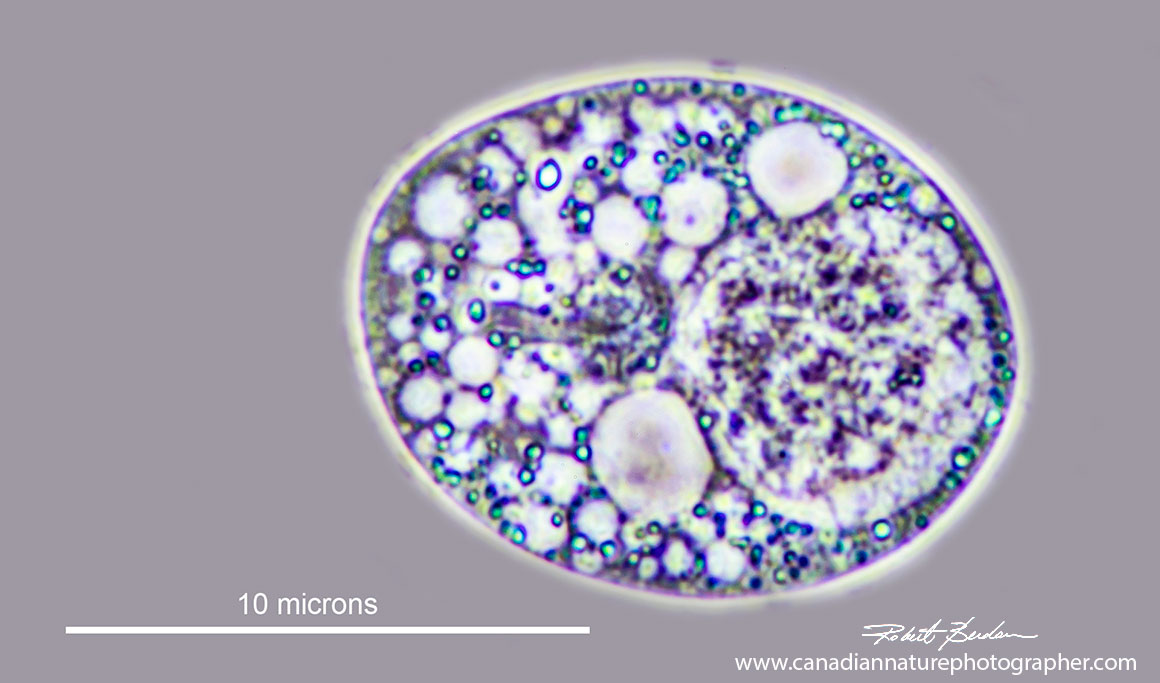
Ciliate found in pond water photographed using positive phase contrast showing intracellular components and external cilia (hair-like structures ) that are used for feeding and propulsion. Zeiss Axioscope Positive Phase contrast 400X.
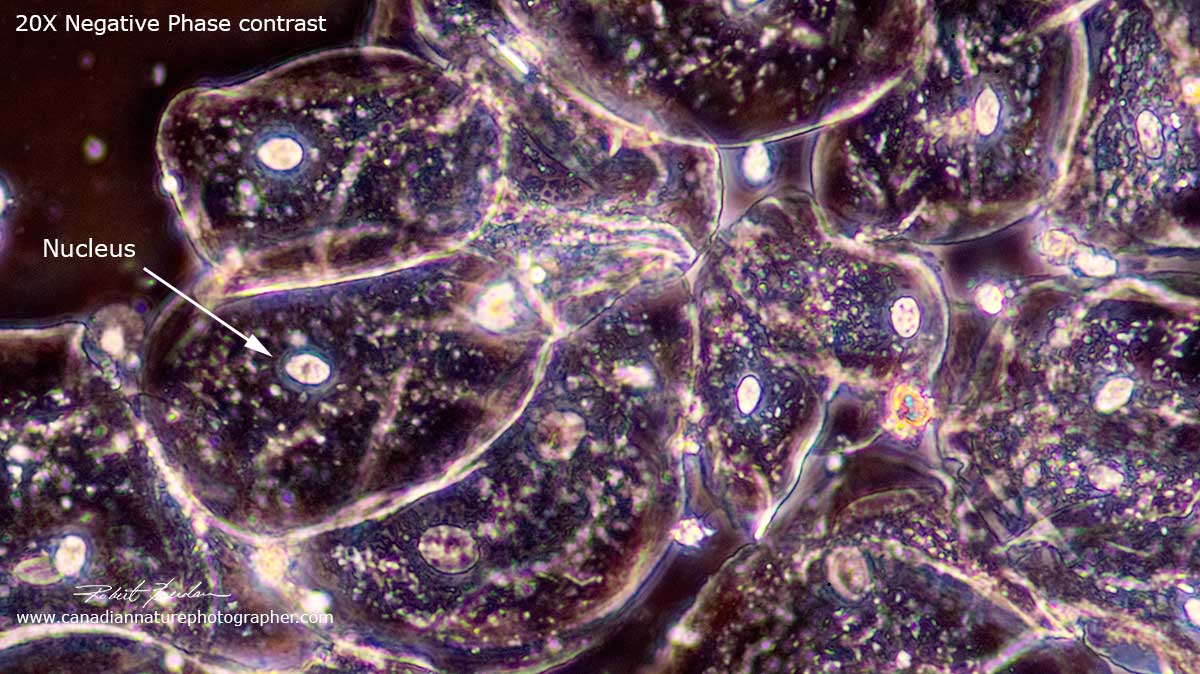
Negative phase contrast image of human cheek epithelial cells showing bright nuclei 200X (microscope objective used was 20X Plan Achromat).

Positive phase contrast human cheek epithelial cells, nuclei are dark in colour on a light background 100X. Positive phase-contrast is the most common type sold.
Polarized Light Microscopy
A polarizing microscope can also produce contrast provided the specimens or cellular components are birefringent (doubly refracting). The addition of linear polarizing filters to a bright field microscope is inexpensive. The use of a compensator or wave plate filter can also introduce colors to the subject and background and a polarizing microscope can determine refractive index of mineral or biological specimens if the specimen thickness is known.

Potato starch grains viewed with a polarized light microscope using a quartz wedge compensator 200X (Motic BA310 microscope)

Tardigrade Milenisium tardagradum photographed with polarized light microscopy showing the skeletal muscle fibres (yellow). Birefringent material inside the mid-gut appears granular and the muscles in the buccal pharyngeal apparatus appear bright blue (top left) - 200X Polarizing microscopy with full wave 550nm retardation filter.
Fluorescence microscopy is another way to add contrast in microscopy. An epi-fluorescence illuminator can cost $5,000 or more for an existing microscope (e.g. Motic 2021). The trend is toward using narrow wavelength LED light sources but their cost at present is still high. This technique requires specimens stained with fluorescent dyes, fluorescent labelled antibodies or other fluorescent molecular probes. Depending on the fluorescent probe a long exposure by the light source may kill the cells if UV radiation is used to excite the dye and it can limit the time of observation due to the dyes toxicity and photo-bleaching. Fluorescent microscopes are expensive costing $5,000 or more. My fluorescence microscope uses a mercury-vapour light bulb that costs about $200 and only lasts for about 200 hours. Light emitting diodes (LED) light sources have advantages in that they are cooler, last longer, and are easier to use. The mercury vapour lamps if used too long (more then 200 hours before changing) becomes explosive and can potentially spew toxic mercury. Also I should mention that BG-12 filters attached to a LED flash light or UV LED light can be used to excite some dyes and\or autofluorescence components for use with a both a stereoscope or light microscope and costs significantly less money.
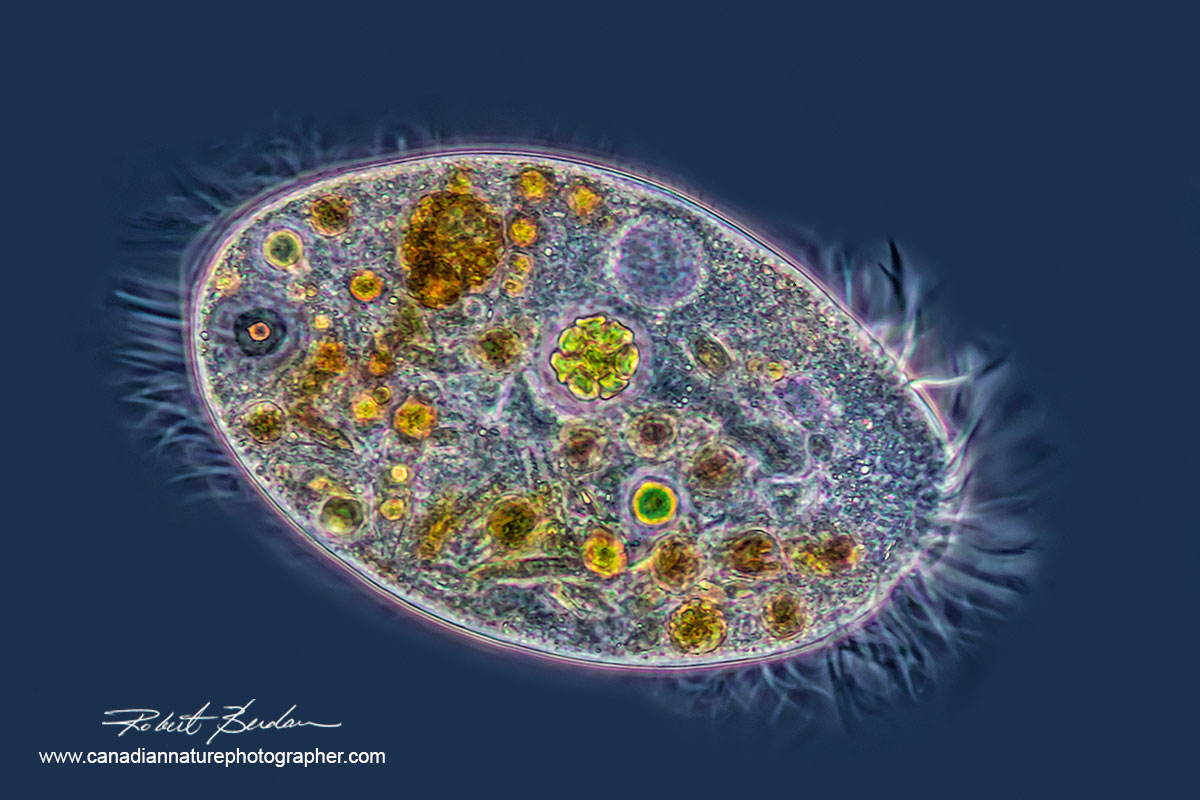
Stentor coeruleus a common single celled ciliate found in pond water is stained with the fluorescent dye Acridine orange 100X. The macronucleus containing DNA stains green and appears like beads-on-a-string in this organism. Acridine orange stains mRNA red and lysosomes (digestive vacuoles) orange-red if the lysosomes have an acidic pH.
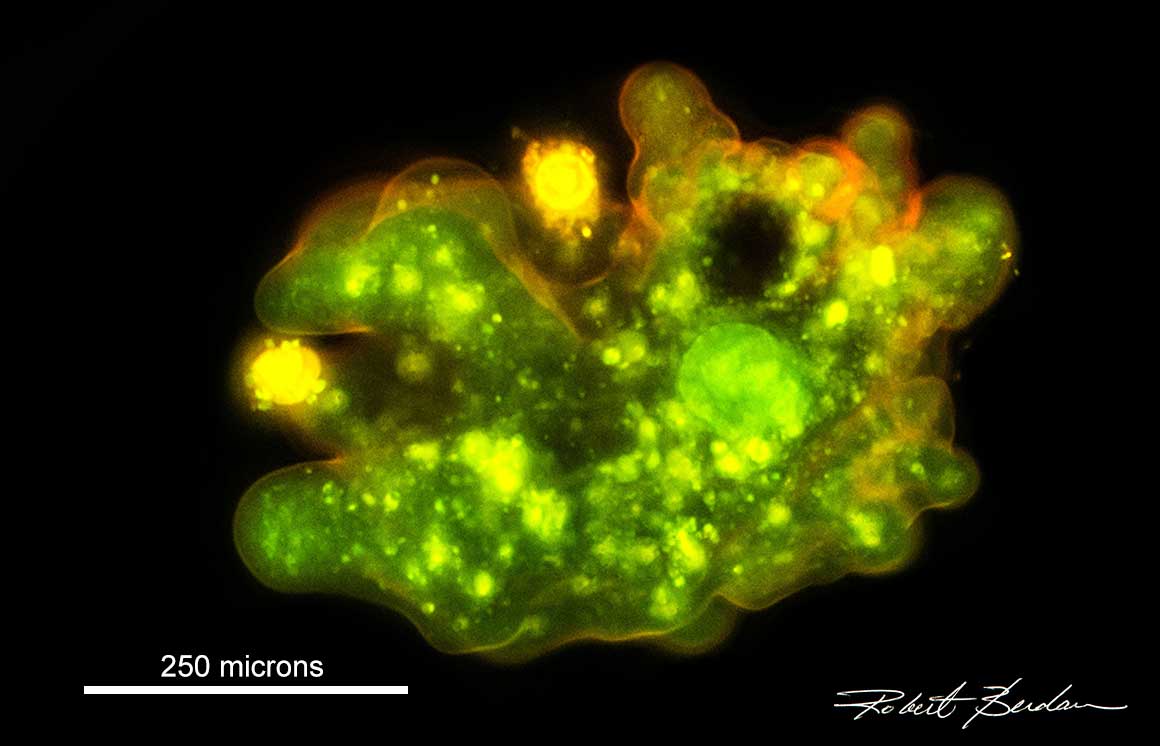
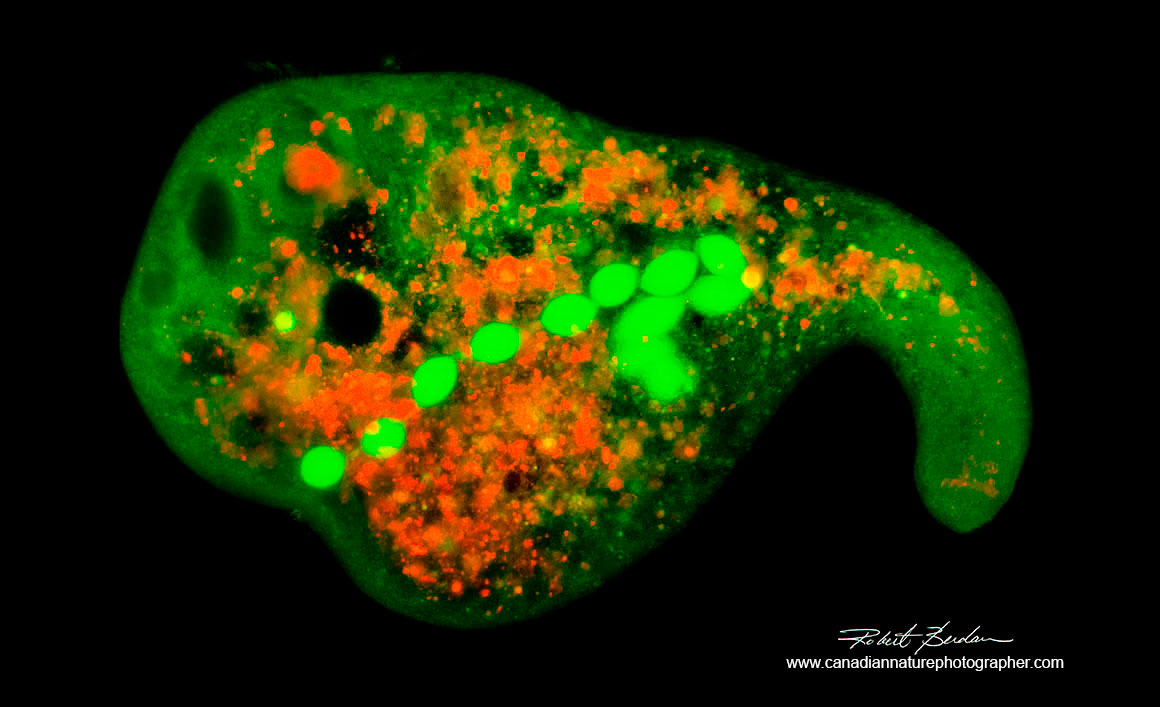
Amoeba proteus Flourescence microscopy Acridine orange stain - 200X.
Differential Interference Contrast (DIC) Microscopy
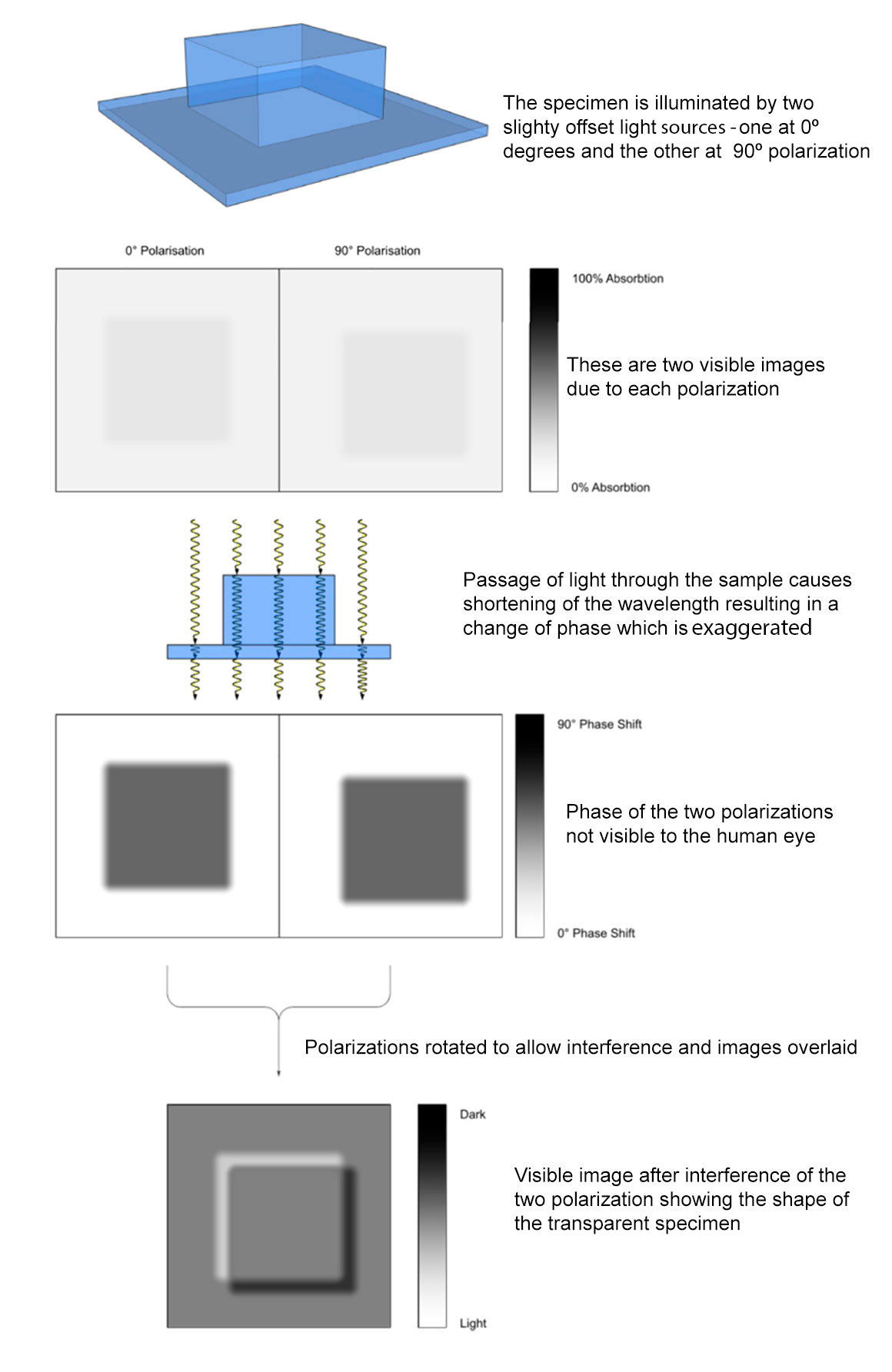
Differential Interference contrast (DIC) microscopy was invented by Francis Smith in 1947 and further developed by Georges Nomarski in the 1950’s (C.R. Bagnall, 2012, M. Shribak, 2012). DIC is sometimes referred to as Nomarski microscopy. DIC works by separating a polarized light source into two orthogonally polarized beams (by a Woollaston prism) which are separated (sheared) a small distance apart. The distance the beams are separated by the first Wollaston prism built into the condenser is smaller than the resolution of the microscope objective being used. The light beams are split and then recombined using a second Wollaston prisms at the back of the objective focal plane and analyzer. This optical path difference in each of the two light beams caused by differences in thickness and refractive index of the specimen cause them to to interfere when combined. This produces contrast and makes the cell appear three dimensional as if lit form the side. Using DIC there are no halos around organelles so the resolution is maximized and the technique permits observation of thin optical sections. The production of DIC is complicated and expensive and can cost more than $10,000 to add this technique to a microscope. DIC is available from the major microscope manufacturers (Zeiss, Leica, Nikon and Olympus) and at least one company from China (Optu EDU) that I am aware of.
A used DIC microscope for transmitted illumination on Ebay.com starts at about $6,000 US which puts this technique out of reach of most amateurs. I have owned an Olympus inverted microscope IMT-11 microscope with DIC for research in the past and I currently own a Zeiss Axioscope A1 with DIC used for taking the DIC images used in this article and on my web site. A lower cost alternative technique is Hoffman modulation which offers similar 3D-like images and the use of oblique lighting can produce DIC-like images as shown further below.
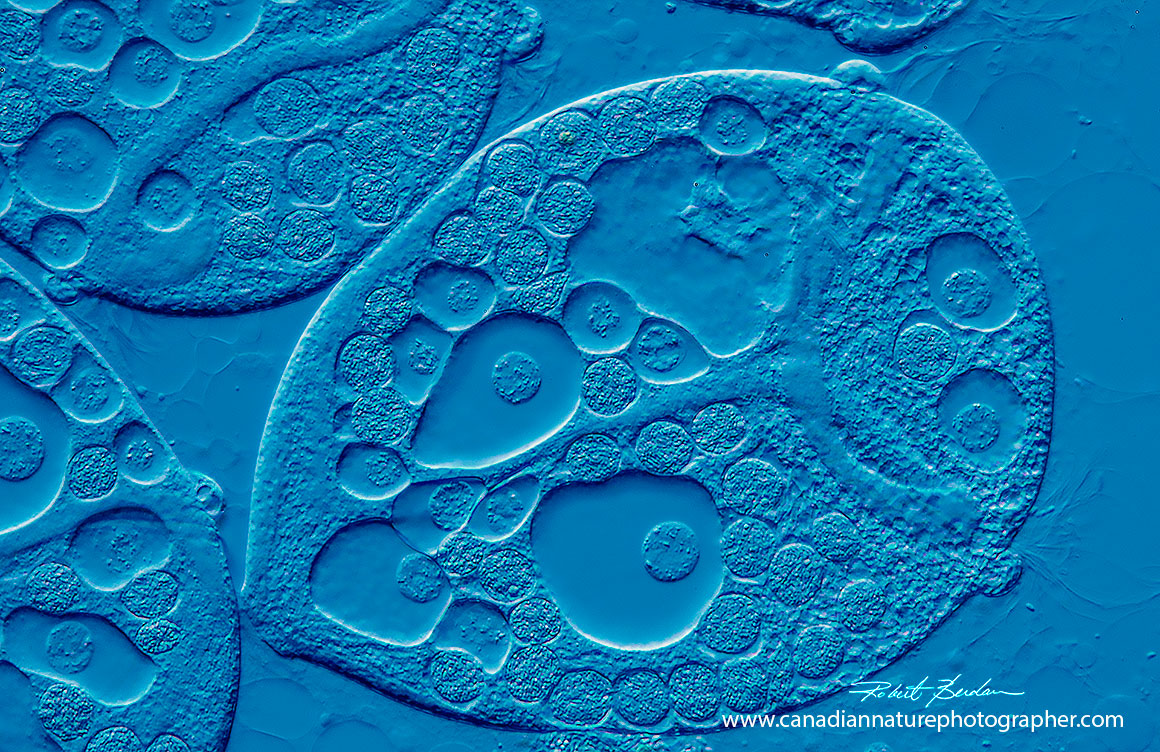
Fresh water Peritrich ciliate by DIC microscopy showing a pseudo 3D-like appearance 400X.
Types of DIC Microscopes
1. Transmitted light DIC used for viewing cells, embryos and other aquatic micro-organisms
2. PlasDIC used for viewing live cells grown in plastic dishes (Hoffman modulation also does this)
3. Epi-scopic DIC for viewing semi-conductors, and other reflective surfaces
Transmitted Light DIC
A DIC microscope uses polarized light and includes a polarizer before the condenser and an analyzer after the objective back focal plane. The use of two polarizers and their consequent reduction in overall light intensity means that DIC microscopes require a bright light source. My Axioscope uses a 100W tungsten bulb. The first Wollaston prism splits the input polarized light into two orthogonally polarized beams with a small shear (separation distance). The split light rays pass through the cell and different refractive indices and thicknesses resulting in differential phase difference. After passing through the objective the second Wollaston prism joins the two beams which undergo interference resulting in dark and light areas seen as amplitude (brightness) differences and greater contrast.
With Smith’s approach to DIC the back focal plane of the objective was located inside the objective which is not easily accessible. Nomarksi modified the second Wollaston prism so it could be positioned after the objective focal plane (M. Shribak, 2012). The second crystal wedge, the Nomarksi prism (modification of the Wollaston prism) recombined the two split beams causing the two rays of light to interfere after passing through the specimen and analyzer. This results in different shades of grey for different regions of the cell depending on its thickness and refractive index. The contrast is produced by the optical path difference in a small in-focus volume where the two interfering beams are spatially separated.
A DIC microscope can be modelled as the superposition of one image over the over image but displaced a small amount and phase shifted by a bias (M. Shribak, 2012). Most DIC microscopes change the optical bias by laterally shifting of the top prism using a small screw (shear control knob) which moves the prism and if a full wave compensator is inserted into the light path, turning the screw will result in different colour backgrounds. This optical staining can be beautiful, but the colours have limited scientific applications (D. B. Murphy et al. Olympus web site).
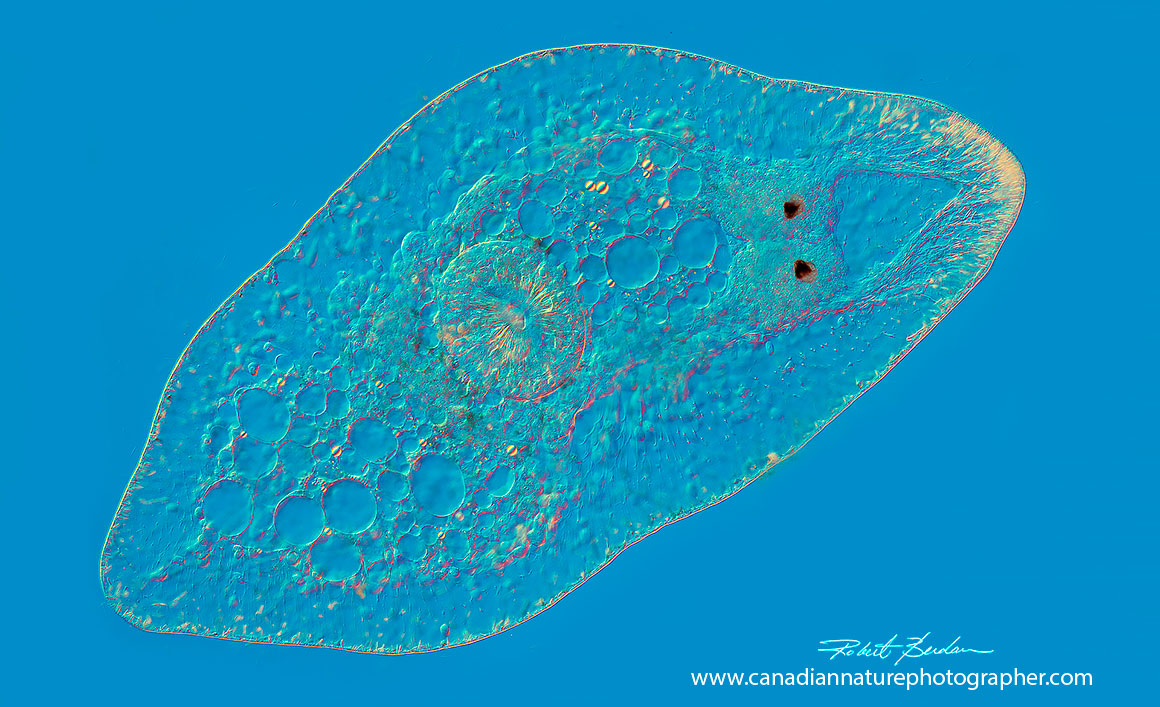
Aquatic flatworm, the two black spots are eyes and the central region shows the opening to the gut. DIC microscopy 50X.
If the specimen contains birefringent properties it may be coloured by DIC due to light interference. A compensator used in a polarizing microscope (full 550 nm) can be inserted in the light path and alter the background colour by interference. The 2nd prism has a shear control that affects the separation of the two light beams and can alter the bias (separation of the light beams) resulting in different shades and tone visible in the background. A DIC microscope is essentially an enhanced polarizing light microscope.
Some researchers recommend using a rotating stage with a DIC microscope because the image gradients need to be aligned at right angles to the direction of shear. That said I own an XY translation mechanical stage on my DIC scope because it is more ergonomically convenient and allows to me to search, find and follow moving aquatic organisms. In 2004 Zeiss launched a C-DIC microscope that uses circular polarized light filters with rotating and sliding prisms so it is possible to depict phase structures regardless of their alignment without having to rotate the subject (R. Rainz and P. Gretcher, 2004).
Transmitted DIC cannot be used to examine cells growing in plastic culture dishes because the plastic is birefringent. One solution I used to do in my research was to grow cells in plastic dishes with glass coverslip bottoms that now can be purchased (e.g. https://www.mattek.com/store/p35g-1-5-14-c-case/). .
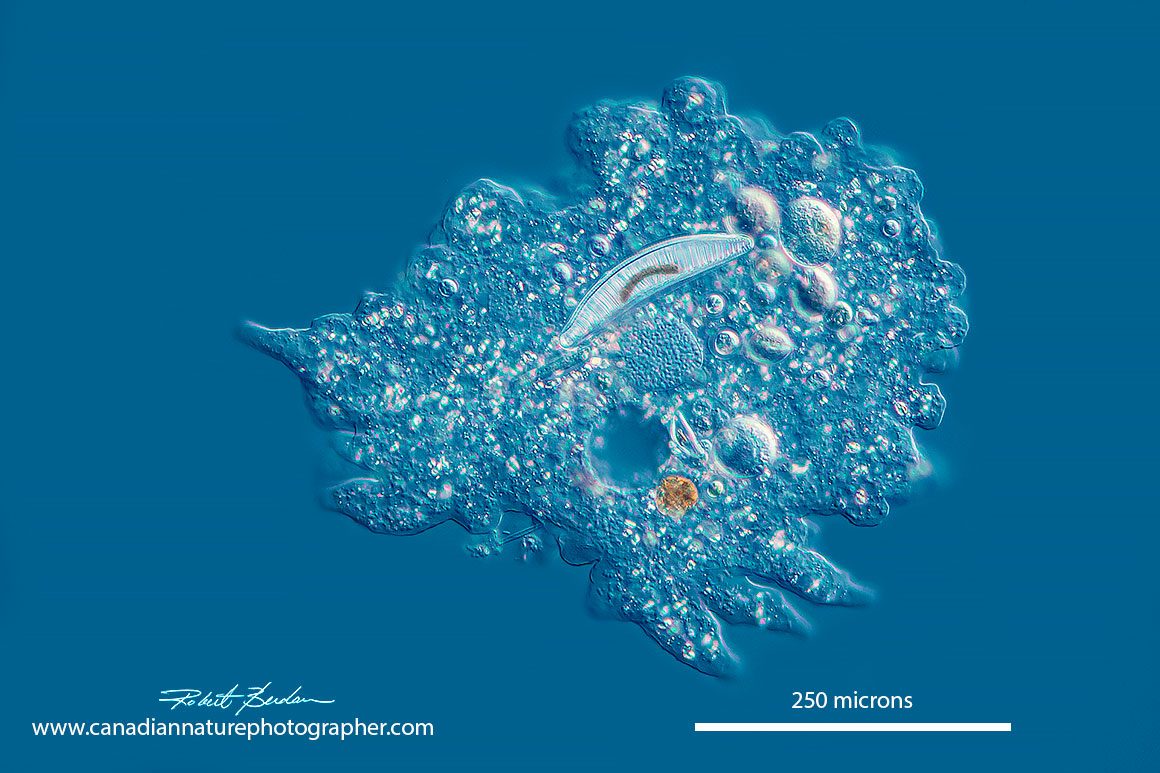
Amoeba proteus by DIC microscopy contains a central nucleus and it has ingested a diatom above the nucleus. Amoeba move slowly yet they seem to attract smaller prey which they capture with their pseudopods and engulf them.

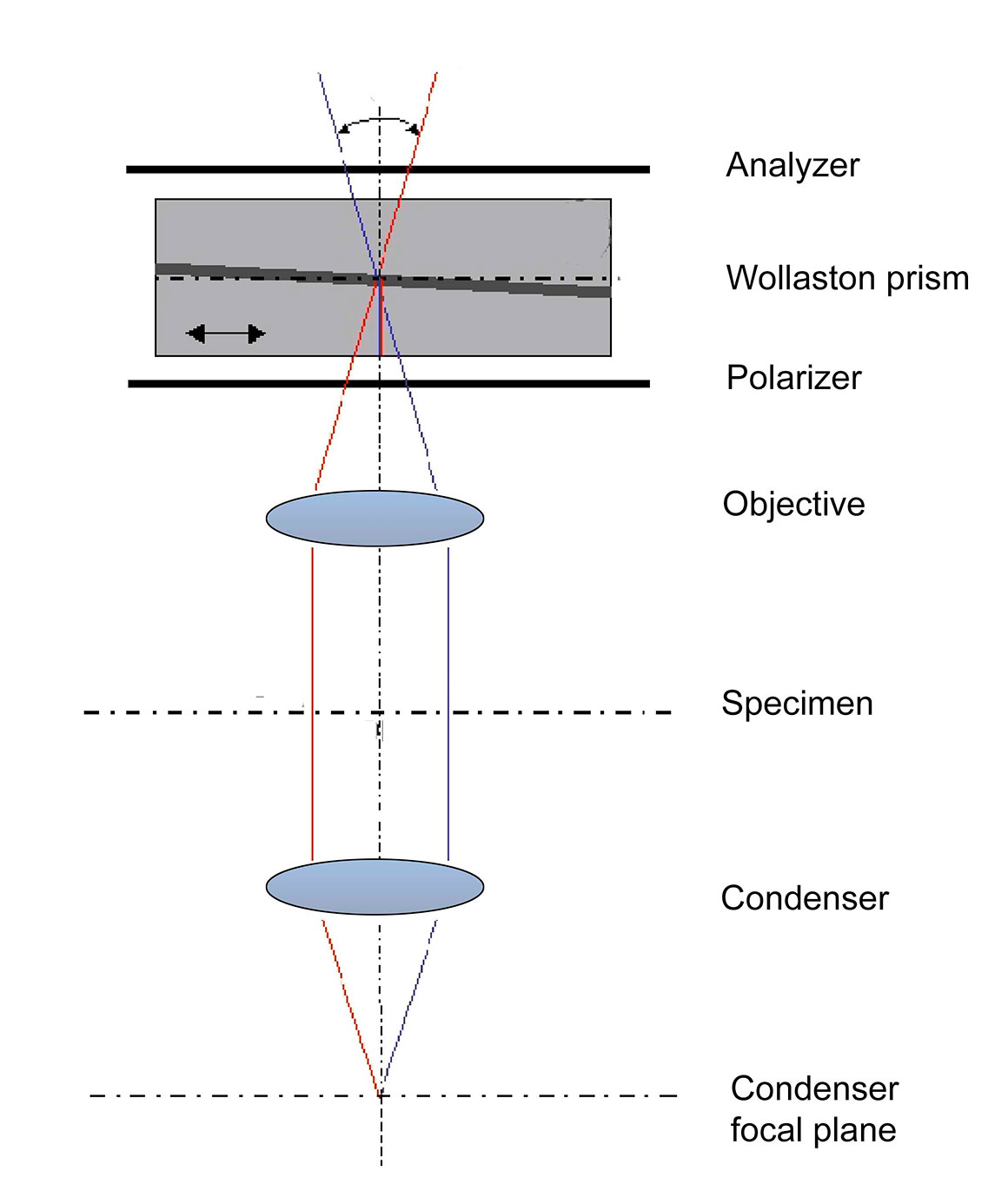
Schematic diagram of a Differential Interference contrast (DIC) microscope.
Diagram modified - the original i by Richard Wheeler (Zephyris) – Wikpedia Public Domain https://commons.wikimedia.org/wiki/File:DIC_Light_Path.png
With DIC microscopy the Nomarski prisms positioned above the objectives in a upright light microscope must be matched with the lower prism in the condenser. The prisms in the condenser are matched to a narrow range of objectives e.g. condenser setting I supports 5X and 10X objective, condenser setting II supports the 20 and 40X objectives, condenser setting III supports 63X and 100X objectives on my Zeiss Axioscope. The prisms are thin and cut with close tolerances; avoid touching them.
Differential Interference Contrast. The light in this diagram is coming from the top down and shows how the 3D image is formed. Diagram modified from Richard Wheeler (Zephyris) Wikipedia.
PlasDIC
PLasDIC is a modification of DIC by Smith and Normarksi and can be used to view live cells growing in plastic culture dishes. PlasDIC was developed by R. Danz, A. Vogelsgand and R. Käthner at Carl Zeiss. The typical relief contrast is produced by the optical path difference (refractive index and thickness) of the specimen compared with its environment. This technique launched onto the market by Carl Zeiss on inverted microscopes in 2003. Each condenser has only one slit diaphragm to produce partially coherent light and it works for 10, 20 and 40X objectives. Where image quality is important regular DIC microscopy using Petri dishes with cover glass bottoms offers higher and better image quality (in normal glass Petri dishes the glass is too thick).
Above is a diagram of Plas DIC showing that uses a single Wallaston prism afer the objective and surrounded by 2 polarizers . This new layout permits DIC imaging with the use of Plastic for growing live cells. Diagram modified from R. Danz, A. Vogelgsang and R. Käthner (2004).
Images taken with Normal Transmitted DIC
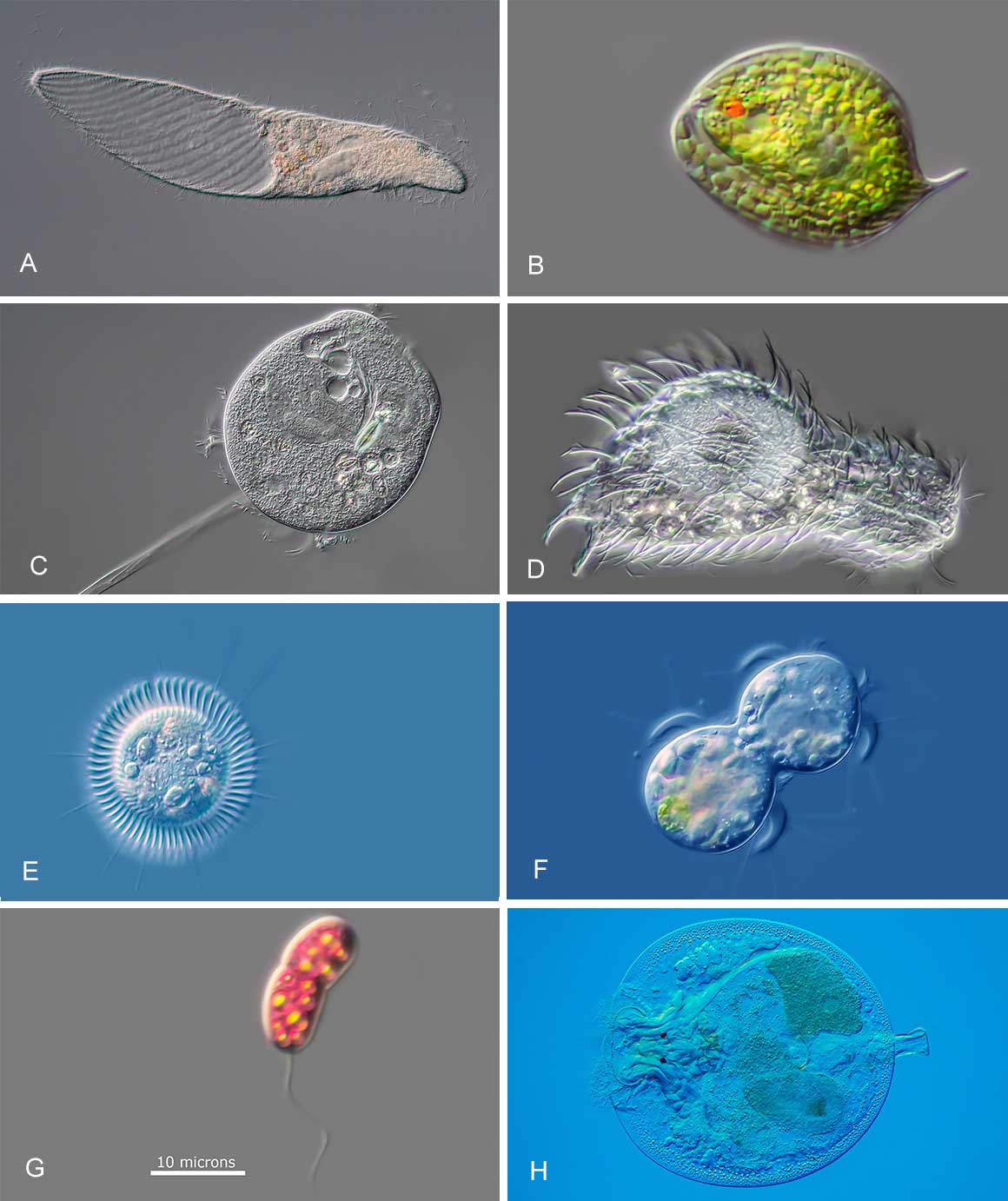
Variety of micro-organisms from a freshwater pond photographed with DIC microscopy. A) Ciliate Spirostomum terres with large water vacuole on left side B) Phacaceae Phacus sp C) Pertrich ciliate D) Gastrotrich E) Ciliate Askenasia viewed from above F) Ciliate Askenasia from the side - undergoing cell division G) Sulphur bacterium H) Rotifer Testudinella patina nicknamed "Turtle Rotifer”. Magnification in each image varies between 200 to 1000X.

Chaoborus (midge fly larvae) swim bladder 200X DIC microscopy

Chyodorus a small Cladoceran living in Fresh water DIC microscopy 100X

Human inner-cheek epithelial cells DIC microscopy 200X
Episcopic DIC or Reflected Light DIC microscopy
When searching for used DIC microscopes on E-Bay I sometimes encounter DIC microscopes for sale fot a few thousand dollars and then notice it is an episcopic DIC meaning that the light source comes from above and passes through the objective which also serves as a condenser. A partially reflective mirror directs the light down onto the specimen then directs the reflected light to the eyepieces or camera. These microscopes have only one Wollaston prism and are designed for examination of opaque highly reflective specimens like metals, circuits, silicon wafers and\or metallic surfaces. These microscopes are not suitable for studying living cells or aquatic microorganisms. For those more interested in reflected light DIC microscopy see article and pictures on Nikon Microscopy University (C Brandmaier et. al.) in the reference section.
Hoffman modulation contrast
Another option to view live cells growing on plastic dishes by microscopy is to use Hoffman modulation. This form of contrast improvement was invented in 1975 (R. Hoffman and L. Gross (1975). It consists of a condenser with a slit aperture or polarizer, an objective with different density polarizers inside the back focal plane of the objective. The objecive and condenser must be properly aligned (Olympus setup). The images have a 3D pseudo-relief in black and white. The technique is less costly than normal DIC though I found Hoffman modulation akward to get working properly. Both PlasDIC and Hoffrman modulation can also be used for viewing cells on either plastic dishes or glass slides.

Human mucosal epithelial (cheek) cell photographed with Hoffman modulation microscopy 400X.
Oblique lighting has been used a long time and many microscopists discover it by mistake. By accidently putting the condsener filter holder or phase constrast conderser opening to partly block the light source oblique illumination causes a 3D-like effect. You can implement this technique with light blocking filters sold onEbay or make home made filters (see below). The filters are positioned below the condenser or on the light source. The simplicity and low cost make it attractive for anyone using a light microscope.
I plan to write a more detailed description of oblique lighting in the future. For now if you have a filter holder under your condenser on your microscope place the filter holder so it partly blocks the light path and move it back and forth while viewing a specimen until you see the 3D lighting. You can also purchase or make oblique lighting filters to place below the condenser or on the light source. At least one microscope company (LOMO) used to make oblique light condensers for their microscopes that consist of a variable iris diaphragm that can be inserted into the light path from the side using a screw control.

Filters shown above can be purchased for a few dollars on Ebay.com or home made and can produce blique lighting when placed under the microscope condenser. For best results move the filters around under the condenser until a good 3D light effect is prodced.
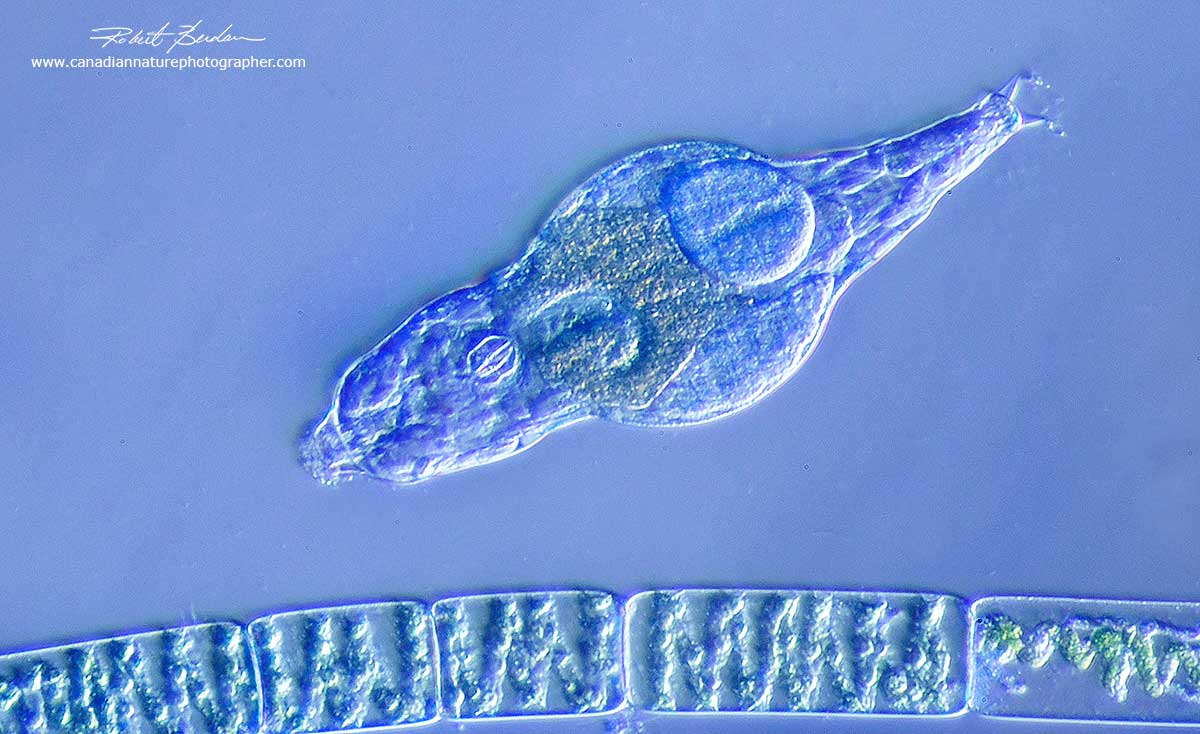
Bdelloid rotifer (top) and Spirogyra algae filment (bottom) of the image viewed in oblique lighting 200X. The main advantage of this technique is it can be produced for under $10.
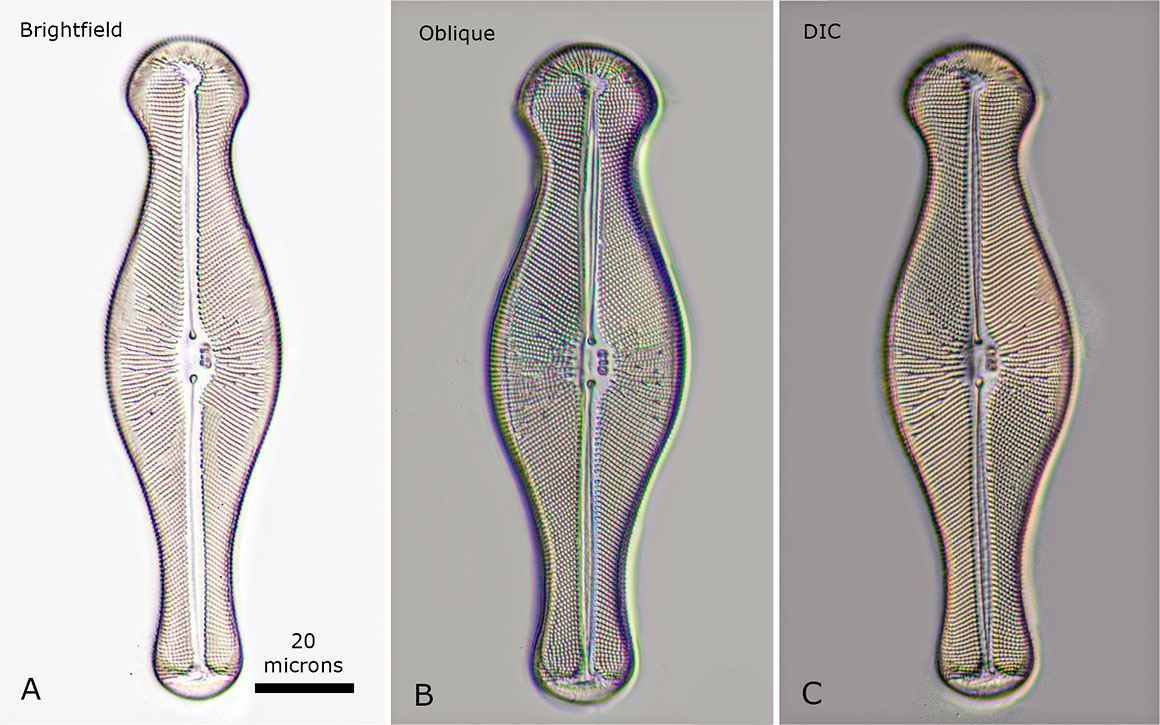
Above is a Diatom Gomphonema sp viewed with three different microscopy techniques. A) bright-field B) oblique lighting and C) DIC microscopy. For diatoms the oblique lighting produces an image similar to the more expensive one produced by DIC microscopy.
Simulated DIC and Phase Contrast - Quantitative Phase Imaging
Software like Photoshop can vastly improve most photographic images and I describe a method (Berdan 2020) to reduce the glow around images taken with a wide-field fluorescent microscope (Photoshop Deconvolution). There are software programs that perform Deconvoluton for confocal microscopy that are better but most are more expensive (Image J\Fijji is free). I came across an article titled Optical Phase Microscopy: Quantitative Imaging and Phase Analogs (B.E. Allman et al, 2002). This paper describes taking three pictures of a specimen with a bright field microscope, stacking the images, one in focus and the two other images taken within the depth of field and then processing them with software using the Transport of Intensity Equation. This computational approach produced images that closely resemble phase contrast and DIC. As of yet I have not been able to get this program. It seems that a software approach is feasible and it could put these techniques into the hands of many more people. Another name for software to simulate Phase contrast is called Quantitative phase microscopy (C.L. Curl et al. 2004) and there are new methods in development that use lensless imaging. For example one study used a birefringent crystal with lensless digital holography to produce DIC images of white blood cells and a nematode (C. Oh et al. 2010). Another science paper describes a method of Quantitative phase-amplitude microscopy to simulate DIC microscopy at a resolution similar to an optical microscope (E.D. Barone-Nugent et. al. 2002, Delbridge et al. 2002). Lens free digital microscopy and other methods offer exciting new developments for microscopy. In the future if a software solution becomes available I will share this information.
Differential Inteference microscopy has been available for over 50 years, but its complexity and cost still make it expensive. Newer super-resoluton microscopes and confocal laser microscopes in research now exceed hundreds of thouscands of dollars. DIC microscopy however, is still expensive for small research labs and amateur microscopists. DIC results in high resolution images that are capable of optical sectioning and are often used in conjunction with other techniques like fluorescence microscopy. Other microscopy techniques including: dark-field, polarization microscopy, phase-contrast, fluorescence and Hoffman modulation are more affordable. Each technique provides different ways to see and examine specimens and micro-organisms. Observing specimens with different microscope techniques provides alternative information about the organism or cells. Digital image processing can also be helpful in improving the clarity of the images and it is possible that in the future we may see more microscopes using a combination of digital sensors and software. I have always been excited by looking through the microscope and the variety of techniques today make the observation of the micro-world even more interesting. The microscope is not just a research tool but it can inspire students, amateurs and artists some of whom may enter exciting fields in biology.
Note: Educators and students may use my web images freely for reports and teaching, all other uses or for commercial use please contact me. If you use my images I appreciate attribution and a link back to this web site if possible. The products mentioned and microscopes brands are those that I have purchased and have worked well for me. All my images can be purchased for prints, advertising etc - please see my image purchase page or contact me by email.
Acknowledgments: I thank Brandon Berdan, Mike Codner and Bruce Taylor for reading the manuscript and making suggestions and corrections.
References & Links to Websites
Differential Interference Microscopy - Wikipedia
https://en.wikipedia.org/wiki/Differential_interference_contrast_microscopy
M. Shribak (2012) Differential Interference Microscopy – Chapt 2 in: Biomedical Optical Phase
Microscopy and Nanoscopy. (eds. N.T. Haked, Z. Salevsky and L.L. Satterwithe), Elseveir.
R. Berdan (2021) Phase contrast by Motic.
https://moticmicroscopes.com/blogs/articles/phase-contrast-by-motic
Motic Fluorescence attachment for Light Microscope
https://moticmicroscopes.com/products/complete-epi-fluorescence-attachment-for-ba?_pos=1&_sid=12af01316&_ss=r
C. Robert Bagnall (2012) Differential Interference Contrast Microscopy – Chapt 11. Pathology 464 – Light microscopy.
https://www.med.unc.edu/microscopy/wp-content/uploads/sites/742/2018/06/lm-ch-11-dic.pdf
D. B. Murphy, K.R. Spring, M. Parry-Hill, M.W. Davidson - Olympus web site
https://www.olympus-lifescience.com/en/microscope-resource/primer/java/dic/opticalstaining/
Hoffman Modulation Contrast Basics – Olympus web site
https://www.olympus-lifescience.com/en/microscope-resource/primer/techniques/hoffman/
R. Danz, A. Vogelgsang and R. Käthner (2004) PlasDIC – a useful modification of the differential interference contrast according to Smith/Normarki in transmitted light arrangement. http://www.zebrasc.com/UpImages/200912/P20091224132510.pdf
R. Hoffman and L. Gross (1975) Modulation Contrast Microscope. Applied Optics 14: 1169-1176.
https://www.osapublishing.org/ao/abstract.cfm?uri=ao-14-5-1169
M.W. Abramowitz and M.W. Davidson Hoffman Modulation Contrast Basics
https://www.olympus-lifescience.com/en/microscope-resource/primer/techniques/hoffman/
Hoffman Modulation Contrast Microscopy - Wikipedia
https://en.wikipedia.org/wiki/Hoffman_modulation_contrast_microscopy
D. B. Murphy, E. D. Salmon, K.R. Spring, M. Abramowitz and M.W. Davidson – Fundamental Concepts in DIC Microscopy.
https://www.olympus-lifescience.com/en/microscope-resource/primer/techniques/dic/dicintro/
C. Brandmaier, K.R. Spring and M.W. Davidson Reflected Light DIC microscopy
https://www.microscopyu.com/techniques/dic/reflected-light-dic-microscopy#
R. Danz and P. Gretscher (2004) Thin Solid Films 462-463: 257-262. Elesvier
http://www.focuspi.com/Site/C-DIC_files/smpolmic_e.pdf
J. Rheinberg (1896) Notes on Colured Illumination. J. of the Royal Microscopical Society pp 373-388.
http://www.mccroneinstitute.org/uploads/Havics_RheinbergIllumination_62-4_pp157-169_2014-1614882343.pdf
R. Berdan (2017) Rheinberg Filters for Photomicrography
https://www.canadiannaturephotographer.com/rberdan_Rheinberg_filters2017.html
A.A. Havics (2014) Contrast Methods in Microscopy: Rheinberg Illumination. The Microscope 62; 157-169.
http://www.mccroneinstitute.org/uploads/Havics_RheinbergIllumination_62-4_pp157-169_2014-1614882343.pdf
C.L. Curl, C.J. Bellair, P.J.Harris, B.E.Allman, A. Roberts, K.A. Nugent and L.MD Delbridge (2004 ) Quantitative Phase microscopy – a new tool for investigating the structure and function of unstained live cells. Proceedings Australian Physiological and Pharma logical Society 34: 121-127.
http://aups.org.au/Proceedings/34/issue.pdf
Rheinberg Filter sets available on Ebay
https://www.ebay.com/itm/274295172218
M. Shaw (2011) How to Make Rheinberg Filters. Micscape Magazine
http://www.microscopy-uk.org.uk/mag/indexmag.html?http://www.microscopy-uk.org.uk/mag/artdec11/ms-Rheinberg.html
C. Sanchez et al.( 2017) Oblique Illumination in microscopy: A quantatitive evaluation Micron.
https://www.sciencedirect.com/science/article/pii/S0968432817303888?casa_token=z_bj3T47-qgAAAAA:RRFqKbNb722PBSO6aPaWlUX36q3tO5UYMYnTfewyGDK_vXBMFllllQV5YGfspoevV4xFpfntUQ
J.R. Baker (1948) A new Method for Oblique Microscopical Illumination Q J Microsc Sci 1948 89 (Pt 2):233.
C. Oh, S. O, Isikman, B. Kahdemhossinidh, and A. Ozcan (210) On-chip differential interference contrast microscopy using lensless digital holography. OPTICS EXPRESS 18:
https://www.osapublishing.org/oe/fulltext.cfm?uri=oe-18-5-4717&id=196029
E.D. Barone-Nugent, A Barty, K.A. Nugent (2002) Quantitative phase-amplitude microscopy I: optical microscopy. J. of Microscopy 206. 194-203.
https://pubmed.ncbi.nlm.nih.gov/12067363/
L.M.D. Delbridge, A.A. Kabbara, C. Bellaire, B.E. Allman, L. Nassis, A. Roberts, and K.A. Nugent (2002) Quantitative Phase Imaging - a new way to "see" cells. Today's Life Science, March\April 2002.
https://www.researchgate.net/publication/295770944_Quantitative_phase_imaging_-_A_new_way_to_'see'_cells
Authors Biography & Contact Information

Bio: Robert Berdan is a professional nature photographer living in Calgary, AB specializing in nature, wildlife and science photography. Robert retired from Cell\Neurobiology research to pursue photography full time many years ago. Robert offers photo guiding and private instruction in all aspects of nature photography, Adobe Photoshop training, photomicrography and macro-photography. Portrait of Robert by Dr. Sharif Galal showing some examples of Robert's science research in the background.
Email at: rberdan@scienceandart.org
Web sites: www.canadiannaturephotographer.com
www.scienceandart.org
Phone: MST 9 am -7 pm (403) 247-2457.