
Scanning Electron Microscopy - Photography
by Dr. Robert Berdan
July 16, 2017

Most of us don't have access to a scanning electron microscope (SEM), but I was fortunate when I was doing research to be able to take photographs with one. Back in the 1980's the scanning electron microscope only provided Black and White images and live material had to be "fixed" and coated with conducting layer of metal usually gold. This has changed today as some microscopes now permit colour and can even be used with live material (insects) e.g. www.scharfphoto.com/technical-descriptions. David Sharf's scanning electron micrographs of animals and plants are perhaps the best known and most beautiful available and he also contributed to the development of the SEM- check his galleries.
I have always been fascinated by microscopes especially the scanning electron microscope because it could produce 3 D images of specimens at high magnification. Unfortunately the high cost of these microscopes limits its use to research staff and graduate students. The scanning electron microscope uses electrons and magnetic lenses instead of light and allows magnifications up to 100,000X or more and has incredible depth of field. To learn how a Scanning electron microscope works visit Wikipedia.

Hitachi offers a desktop model for about $60,000 which might be affordable for some small research firms or museums, the bigger microscopes cost $200,000 or more. Hitachi offers a desktop version which can provide from 20X to 10,000X magnification. I don't see owning one in my future, but you can get access to an SEM at some Universities for an hourly or daily cost. Perhaps some day they will become more affordable and if I am still around I will surely get one.
My first experience with a scanning electron microscope was at the University of Western in Ontario, where I was studying zoology. My first specimen was an aphid that I captured in the grass near my home. The pictures I took below were part of a class assignment and were recored on 35 mm black and white film.

Scanning electron micrograph of an Aphid approx 100X - photographed on 35 mm Plus-X Pan 100 ISO film. Most scanning electron micrographs are Black and White - colour is often added afterwards in an image editing program like Photoshop.
Closeup up of the Aphid via Scanning Electron Microscopy approx 500X - False coloured with Photoshop. Adding colour i engages viewers and sells more pictures.

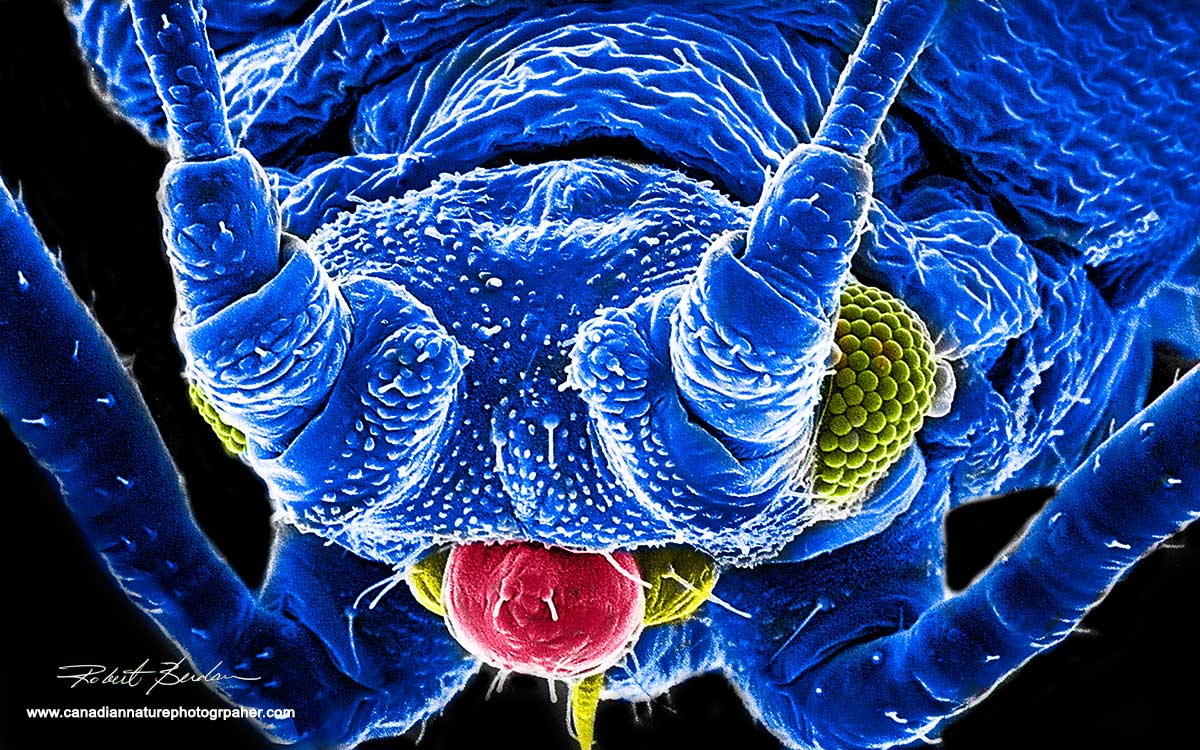
Scanning electron micrograph of an Aphid compound eye and part of his antenna on the left - 1000X

High magnification view of Aphid ommatidia making up the compound eye , there is a single bacterium (pink) resting on one of the facets 10,000X - False coloured.
I have always been fascinated by microscopes of all kinds and spent thousands of hours taking pictures through them. Below are some my scanning electron micrographs which I recorded on film and with a bit of Photoshop processing I have tried to bring them back to life. I worked as a Postdoctoral fellow at the University of Calgary in Neurobiology in the mid 1980's. I was researching how isolated nerve cells from freshwater snails (Helisoma trivolvis) regenerate and form synaptic connections in culture. A picture of the fresh water snail is shown below.

Sun snail Helisoma trivovis is used to study nerve regeneration because their giant brain cells can live at room temperature, in low oxygen and their brain cells (central nervous system) unlike ours, is able to begin to regenerate a few hours after injury. I was trying to understand what triggers this regeneration and what allows them to regenerate so quickly. The snails are red colour because their blood contains hemoglobin like our red blood cells.
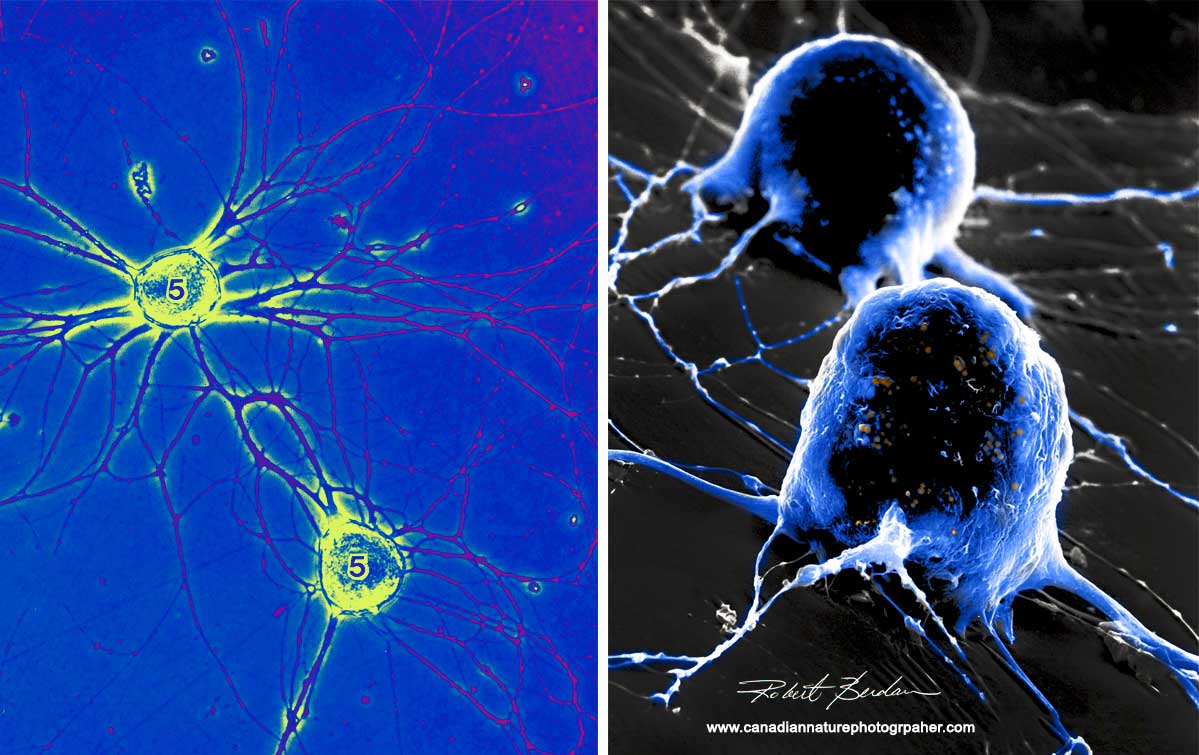
On the left are two isolated snail neurons growing in culture taken with a Phase contrast microscopes (false coloured) 400X. On the right are the same two neurons viewed in a scanning electron microscope which gives a 3D appearance 2000X. The snail has a relatively simple brain and some of the cells and their functions can be identified by their location, appearance and electrophysiological properties. Some of their brain cells can be as large as 1-2 mm in diameter making it relatively easy to extract specific neurons and grow them in culture. One researcher (Dr. Naweed Syed at Univ. Calgary) formed the first simple circuits using these neurons in culture.
I prepared isolated nerve cells for Scanning electron microscopy and one of my photos made it on the cover of the Journal of Neuroscience and another on National Geographic's Web site (see below). I wanted to determine if the isolated nerve cells were free of any attached glia or hemocytes (mollusc's equivalent of white blood cells). I also photographed how the hemocytes interacted with the nerve cells after injury. Below you can see some hemoctyes attached to the nerve cell body and they travelled along outgrowing neurites.

Isolated Neuron from the freshwater snail (Helisoma trivolvis) buccal ganglion grown in cell culture. The neurite extensions are easily seen, the yellow cells are hemocytes. The number is for photographic reference the No. 5 U stands for 5 microns - a micron is 1,000 of a millimetre and this bar can be used to accurately determine the magnification of the pictures. See this picture on National Geographic's web site.
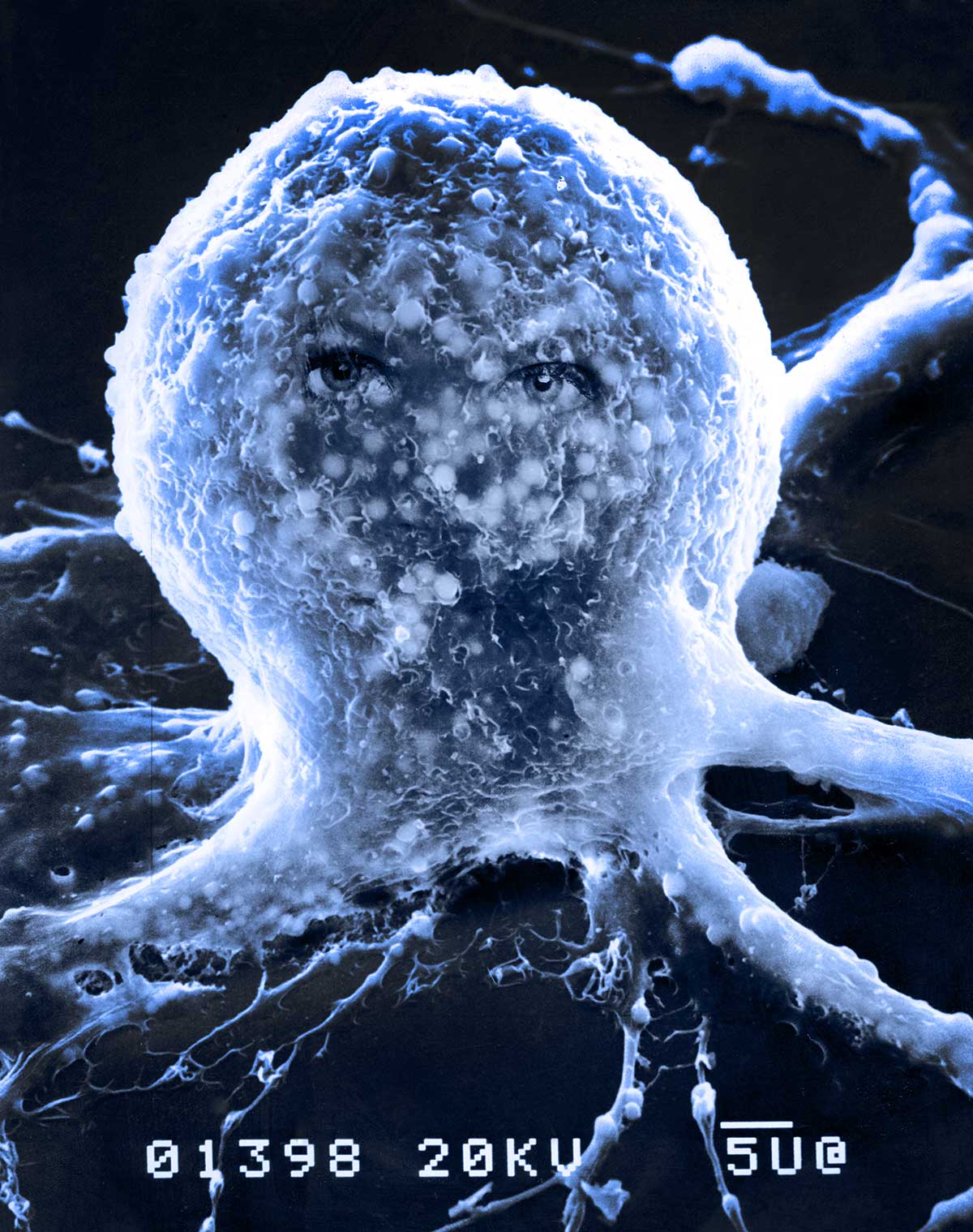
Above is a scanning electron micrograph of an Isolated Neuron (B5) in tissue culture. The eyes of my wife were added in the darkroom before the days of Photoshop and the image is titled "Donna". The neuron made the cover of the Journal of Neuroscience (without the eyes). The small round objects visible inside the membrane are lipid droplets, the electrons penetrate the plasma membrane to show some of the components below. About 2000X . The bar below 5 U bar represents 5 microns. It looks like an octopus to me. Below are the published photos in a science journal.

Above are the photos I presented in research publication Berdan et. al (1990) Journal of Comparative Neurology 296: 437-446 showing the isolated neurons and associated hemocytes. N represents a neurite, the * represents hemocytes or cells associating with the neurons in culture. The small inset (e) show the same hemocyte (d) using a bright field light microscope with phase contrast.
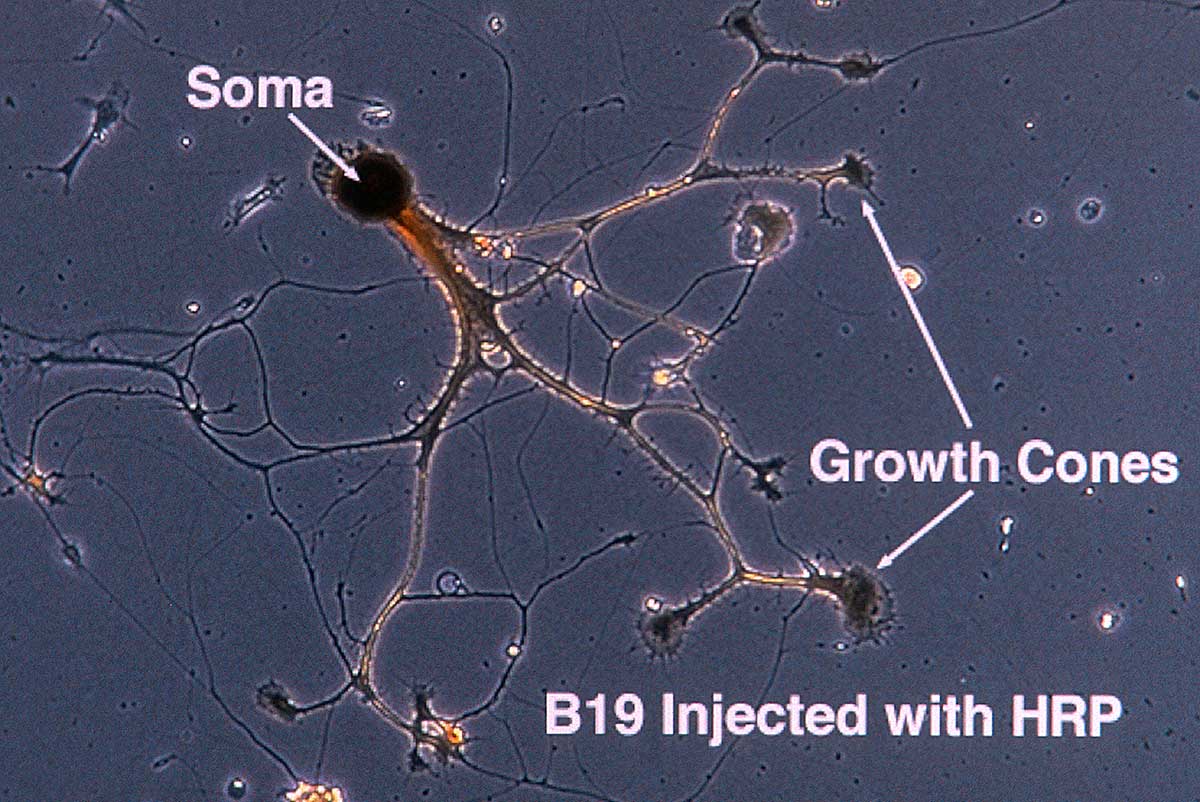
Single Neuron (Soma is the cell body with nucleus) and the growth cones are the "fingers" at the tips of the neurites where the neuron is actively moving and growing. Phase contrast microscopy 400X. The neuron was injected with Horse radish Peroxidase (HRP) so it could be identified in Transmission electron microscopy.

Above photograph shows an isolated neuron using SEM sand the outgrowth of neurites from above. The top right inset shows the tip of one neurons called a growth cone.

Left: Isolated snail neuron placed in a culture dish as viewed with a light microscope (Phase contrast), Right: the same neuron viewed in a scanning electron microscope (bar 5 microns) showing much more surface detail. This is is how the neuron looks when its first isolated from the snail brain - the broken axon retreats into the cell body and then after a few hours begins to grow rapidly. These neurons are very large because they are polyploid - that is they contain several sets of chromosomes and this in part might explain whey they are able to regenerate so quickly.

Hemocytes in close contact with neurites in culture. It's not known whether or not the hemocytes contribute to the ability of the nerve cells to regenerate so quickly. Scanning electron microscopy 2000X
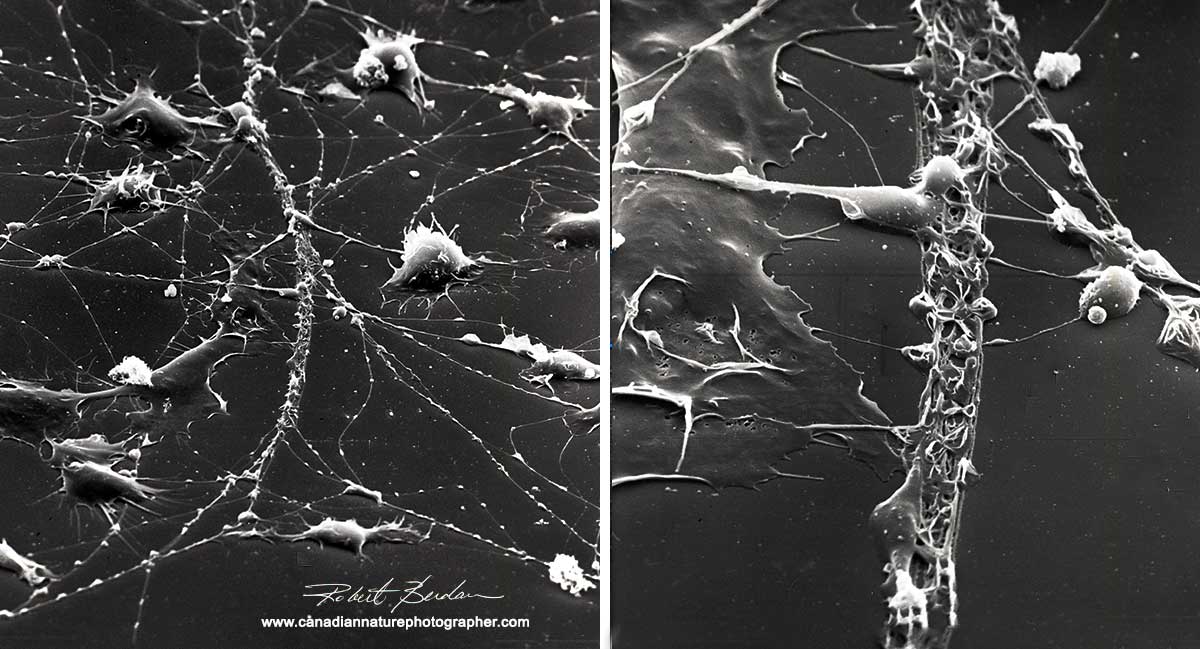
Low magnification views in the scanning electron microscope reveals that many of the cells in culture (hemoctyes) create and follow tracks that appear like slime trails on the dish. Approx 500X.

Left: Hemocyte on the surface of neuron 5000X Right: Closeup of neurite process showing lipid vesicles below the membrane surface (orange spheres) 5000X False colour.

On the tips of growth cones there were small spikes with swellings filled with lipid possibly a reservoir for the formation of new plasma membrane. Bar is 5 microns. False coloured.

SEM of mouse glial cells in culture exposed to cAMP. These cells are far more sensitive to temperature and changes in oxygen levels.

Mosquito proboscis Left: Bright field microscope Right: Scanning electron microscope - false coloured approx. 600X. These pictures are perhaps easier to appreciate as most of us have been bitten by a mosquito - now you can see what they stick into you - ouch!
In the past I recorded images from the scanning electron microscope on 35 mm or 6 x7 cm black and white film. Today digital cameras and computers are used to capture the images. It's an amazing world we live in and when we begin to look more closely we begin to discover interesting things all around us. In a future article I will explore the world of macrophotography using image stacking and show how photographers are capturing arthropods and other animals up close with incredible depth of field - it may be as close as most of us get to owning a scanning electron microscope. RB
References and Additional Links.
Hitachi's Desktop Electron Microscope 2009 priced around $60,000.00 - for home use? Download brochure - PDF
David Scharf's technique for viewing live specimens in the Scanning Electron Microscope
Colorizing Scanning Electron Microscope Images by Ted Kinsman - PDF
National Geographic - Seeing Eye to Microscopic Eye
Pollen photogallery by SEM National Geographic
Want to buy your own scanning electron microscope - me too if I win the lottery
Hemocytes play a role in the immune system of invertebrates - Wikipedia
Also see my other articles on Microscopy:
Rheinberg filters for Photomicrography
The Art & Science of Photomicrography with Polarized Light
Microscopic Life in Ponds and Rainwater
Photographing Through a Microscope Photomicrography - Inner Space
Authors Biography & Contact Information

Robert Berdan is a professional nature photographer living in Calgary, AB specializing in nature, wildlife and science photography. Robert offers photo guiding and private instruction in all aspects of nature photography and Adobe Photoshop training.
Email at: rberdan@scienceandart.org
Web site: www.canadiannaturephotographer.com
Phone: MST 9am -7 pm (403) 247-2457.
Click on the buttons below and share this site with your friends