
Photographing Stentors - A Large Unicellular Ciliate living in Freshwater
by Dr. Robert Berdan
July 10, 2018
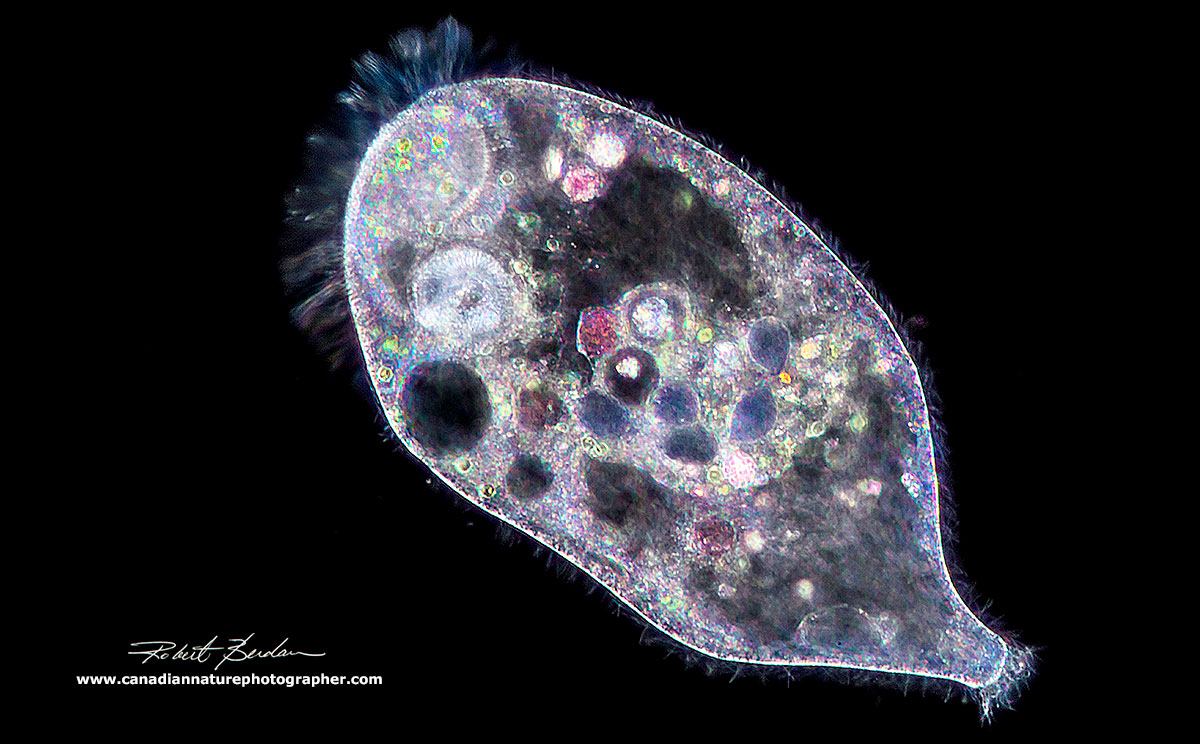
Stentor sp viewed by Darkfield microscopy - this photomicrograph appears abstract and reminds me of Picasso's paintings 200X.
I encountered my first Stentor when I was teenager collecting pond samples to examine with my microscope - see below.
Stentor coreuleus I photographed with an Olympus OM-1 35 mm film camera using my Olympus E microscope with Phase contrast in 1979 on Kodachrome II film - 1\4 and 1\2 sec exposures - over 40 years ago.
In Greek mythology Stentor was a warrior in the Trojan war (13th century BCE) and whose voice was as a loud as 50 men - he encouraged the Greeks to fight. Stentor the ciliate is a microscopic animal and is sometimes referred to as a trumpet animalcule because it can take on the shape of trumpet. Oken in 1815 formally recognized these protists and named them Stentor. For microscopists they are relatively easy to find in ponds as it may be the largest unicellular ciliate reaching a size of up to 4 mm when stretched out. Most Stentors that I have encountered are smaller then this and range from 0.15 mm (150 microns) to about 1.5 mm (1500 microns).
There are 19 Stentor species as of date with only one species found in salt water. Most Stentor species occur in normal or acidic water low in oxygen with high levels of dissolved humus. They are good indicators of organic pollution. Because of their large size they are relatively easy to isolate with an eye dropper. Sometimes Stentors will congregate and attach to an underwater branch or other object and form social groups of several hundred or more.
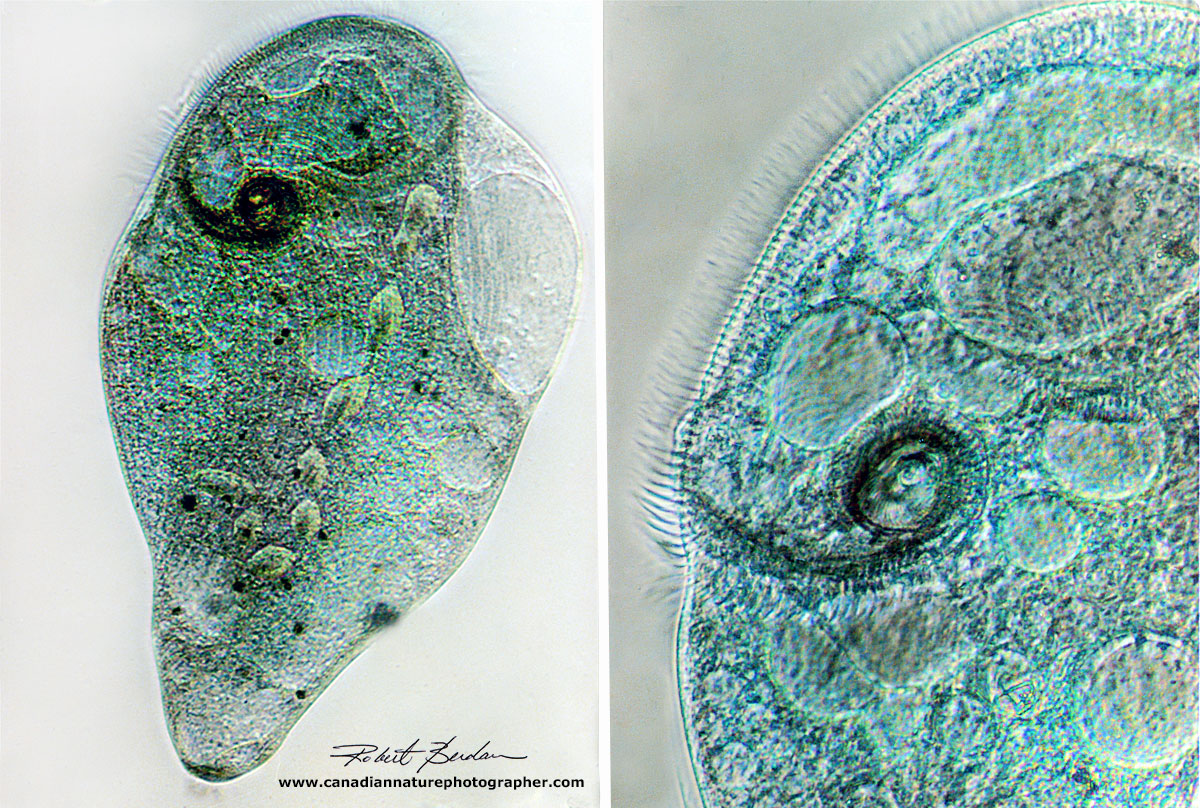
Above is a Stentor coeruleus viewed by Differential Interference Microscopy (DIC) at 200X. The oval beads inside the animal are part of the macronucleus. At the right you can see the membranelles (long cilia or hairs) that lead to the mouth (cytosome) and smaller cilia around the edges of the ciliate.

Stentor viewed by bright field microscopy showing its trumpet form shape and stripes.

Stentor sp viewed by Darkfield microscopy showing endosymbionts - algae living inside.
Stentors can swim freely using cilia or they can attach by their hold-fast on the posterior end and become sessile temporarily. The body is highly contractile and can form into the shape of a ball or stretch out into a long trumpet shape (see below). Cilia covers most of their body but adjacent to the oral apparatus on the anterior end the cilia form an adoral zone of membranelles formed by the fusion of several cilia to form "super cilia". These giant cilia form a spiral that leads into a small buccal cavity which in turn leads to a cytosome (mouth) where food is ingested.

Stretched out Stentor resembling a trumpet - some Stentors can reach 2-4 mm in length. DIC microscopy 100X

Above photo shows high magnification image of the Stentor hold fast that allows them to attach to the substratum. 400X DIC microscopy.

When swimming freely Stentors are often in the shape of a trumpet or barrel, but when they touch something they can quickly retract into a ball shape (top right). 200X DIC microscopy.
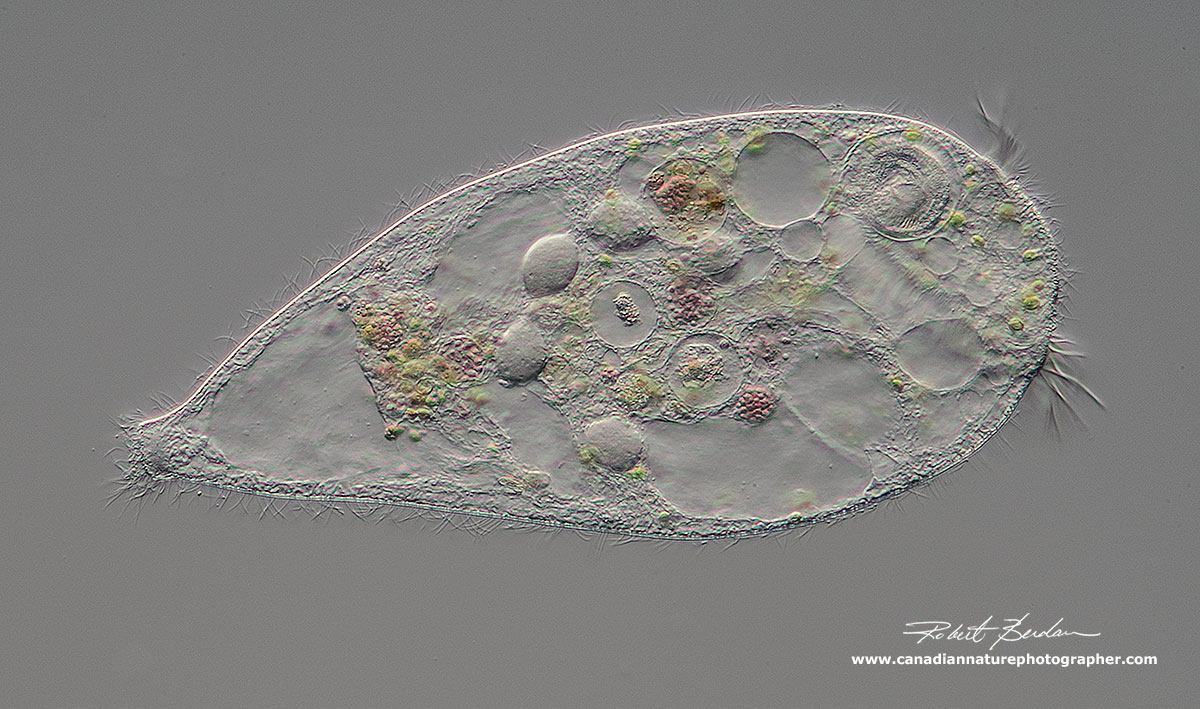
Stentor by darkfield microscopy, anterior end is on the left and the posterior hold-fast is on the right. Darkfield reveals many vacuoles inside the animal, some are clear (dark) and others filled with food. At least one of the clear vacuoles is a contractile vacuole that plays a role in osmoregulation and excretes excess water. 100X.

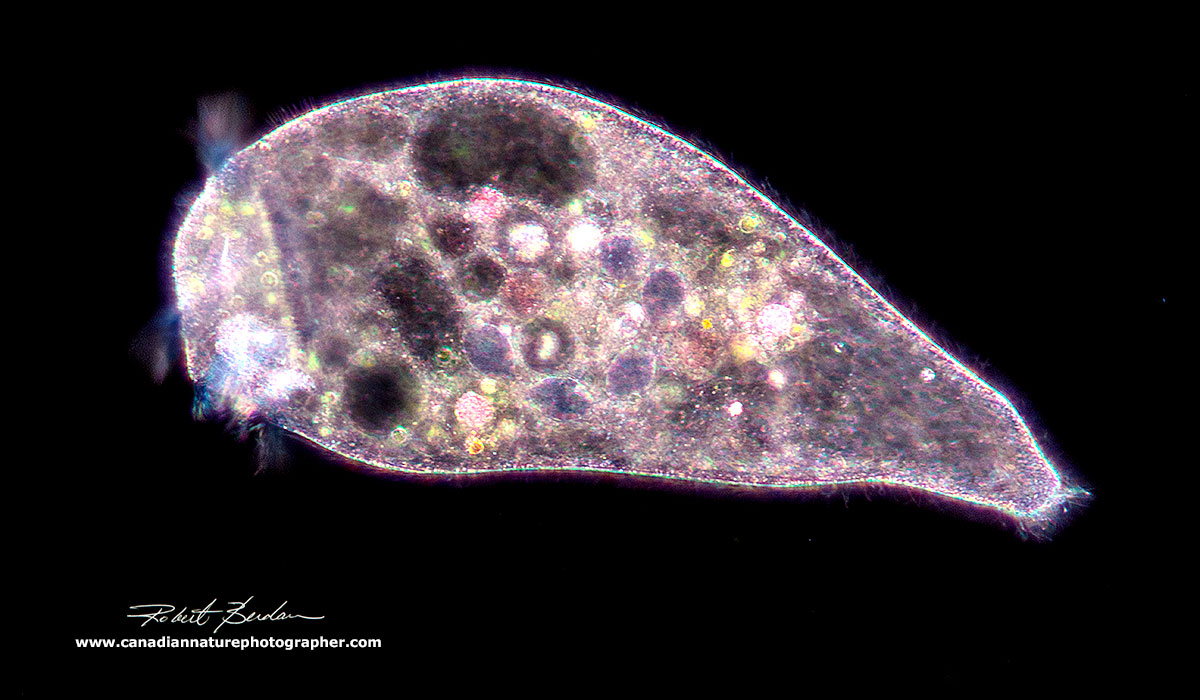
Stentor sp viewed by Darkfield microscopy 100X

Stentor amethystinus with red cortical granules and endosymbiont algae photographed by Darkfield microscopy 100X
Currently there are 19 species of Stentor and the main taxonomic characteristics that differentiate them are 1) presence or absence of 4-7 micron endosymbiotic algae belonging to the Chlorella vulgaris group 2) Nuclear structure and 3) presence and colour of cortical pigment granules (red-purple, green, blue or brown). For identification it is essential to study live cells because the colour of the cortical granules may change after fixation.
Stentor Taxonomic Classification
Kingdom: Chromista - eukaryotic kingdom
Phylum: Cilophora - presence of hair-like organelles called cilia
Subphylum: Postciliodesmatophora - stacks of microtubular ribbons assoc. with kinetosomes
Class: Hetotrichea - prominent zone of membranelles, shorter cilia on rest of body
Order: Heterotrichida - They typically have a prominent adoral zone of membranelles circling the mouth, used in locomotion and feeding, and shorter cilia on the rest of the body. Many species are highly contractile, and are typically compressed or conical in form.
Family: Stentoridae - very large ciliates 0.5 to 4 mm
Genus: Stentor - 19 species

Above photomicrograph of the anterior end of a Stentor as viewed by Bright-field microscopy showing red-purple cortical granules in rows and symbiotic green algae inside this animal. 300X Stentor amethystinus.
The function of the cortical pigment granules is not known. Stentor coeruleus granules contain a pigment called 'stentorin' and its hypothesized that they may mediate orientation of the Stentor in a light gradient and may also offer protection from predation as the substance is photodynamically active and can kill cells that have ingested it when exposed to intense visible light. In Stentor amethystinus the pigment that makes up the cortical granues has been isolated and is called Amethystin (Höfle et al. 2014).

The cortex (pellicle) of Stentor has distinct striae, broad stripes of granules which alternate with narrow clear furrows containing the ciliary rows. Pigment granules (0.5 - 2 microns) can be red, blue, green or brown in colour. Light microscopy 400X. Stentor amethystinus

Above Stentor coreuleus with green cortical granules forming M bands and you can see the clear areas (Km bands) on the surface of this specimen. DIC microscopy 100X (animal from Boreal science).
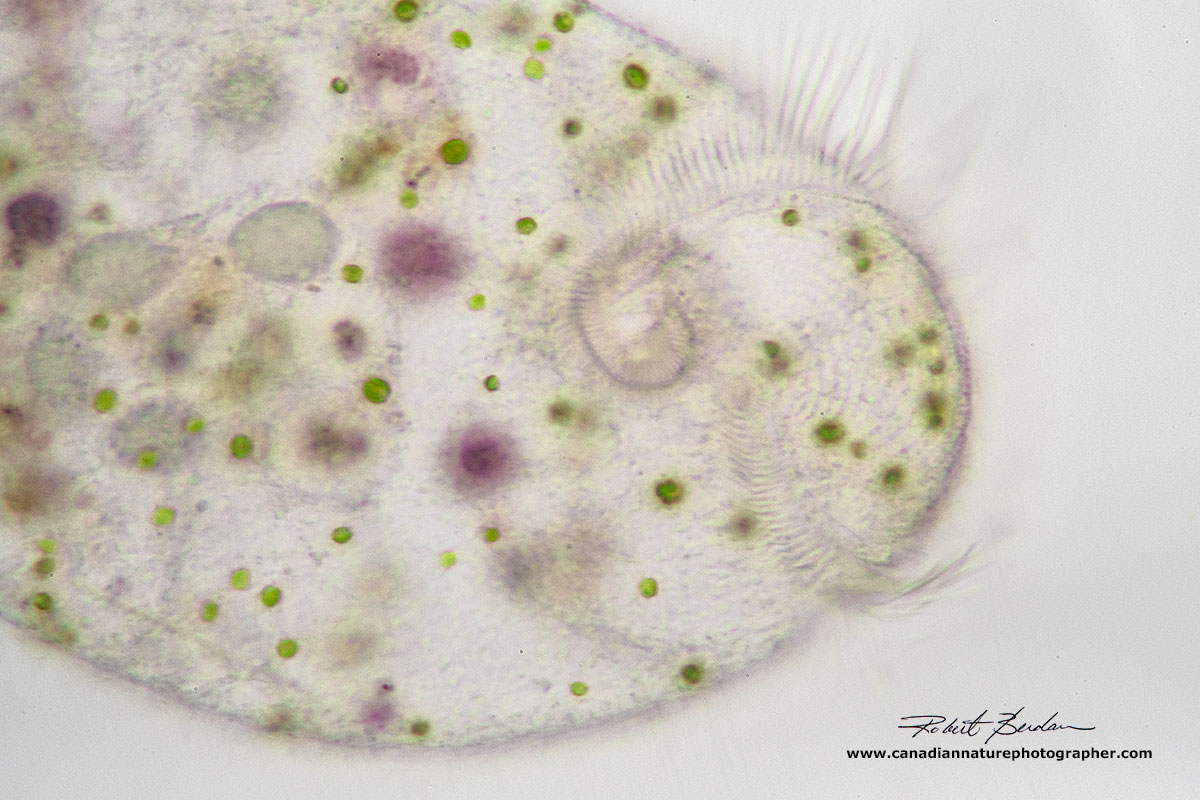
Brightfield microscopy of Stentor showing green symbiotic algae inside. The algae provide the Stentor with food (sugar) and oxygen, whereas Stentor provides the algae with carbon dioxide, nitrogen and other nutrients. Stentors with symbiotic algae show positive photo-taxis (move toward the light). Brightfield microscopy. 100X

On the left are the membranelles that form a circle leading to the cytosome. On the right you can see the "beads" on a string (monoliform) macronucleus. DIC microscopy 400X

The same view of the Stentor shown immediately above, but by using Darkfield microscopy. 400X Endoplasmic vacuoles give the Stentor cytoplasm a spongy appearance.
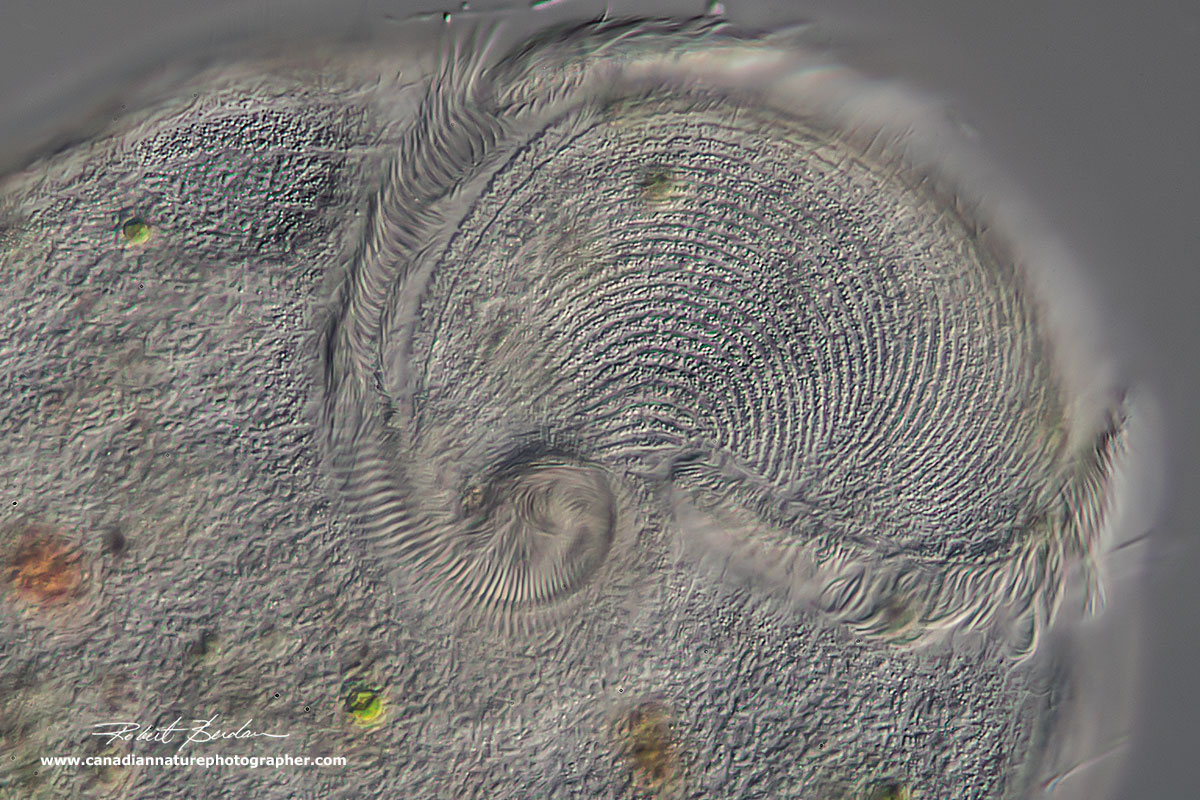
This is DIC microscope image of the Oral zone of another Stentor showing how it spirals into the cytosome. DIC microscopy 400X

Stentor viewed by Polarized light microscopy, the chlorella inside appear bright yellow. 200X

Polarized light micrography of Stentor, negative image 200X.
In the image above I reversed the image to show the Km fibers (dark stripes) more clearly. Some of the fibers have lateral branches (white). The wider spaces are the M bands where cortical granules are found. Studies by electron microscopy (Huang and Pitelk 1973) show the Km fibers are made up of microtubles with thin filaments below them and are believed to produce the contractions in the ciliate. Calcium is believed to be important in the contraction process just as it is in mammalian muscle.
Stentor Macronucleus
The macronucleus of Stentor can take on many shapes - see below. It can be vermiform, nodular or monilform with regular spaced chain of beads connected by thread like elongation of the the nuclear membrane. The macronucleus is polyploid meaning it contains many copies of each gene. The Macronucleus is surrounded by one or more micronuclei but with out special DNA staining the micronuclei can be difficult to see.

Stentor macronucleus diagram from W. Foissner and S. Wölfl (1994)

Stentor muelleri with a moniliform macronucleus with 12 beads. DIC microscopy 100X

Stentor muelleri showing a moniliform macronucleus 200X

In the above pictures it's possible to see the beads of the macronucleus (moniliform macronucleus) and the string like attachments made up of nuclear membrane. You can also see some of the symbiotic algae in the cytoplasm. DIC microscopy of extruded cytoplasm 600X. Stentor muelleri.

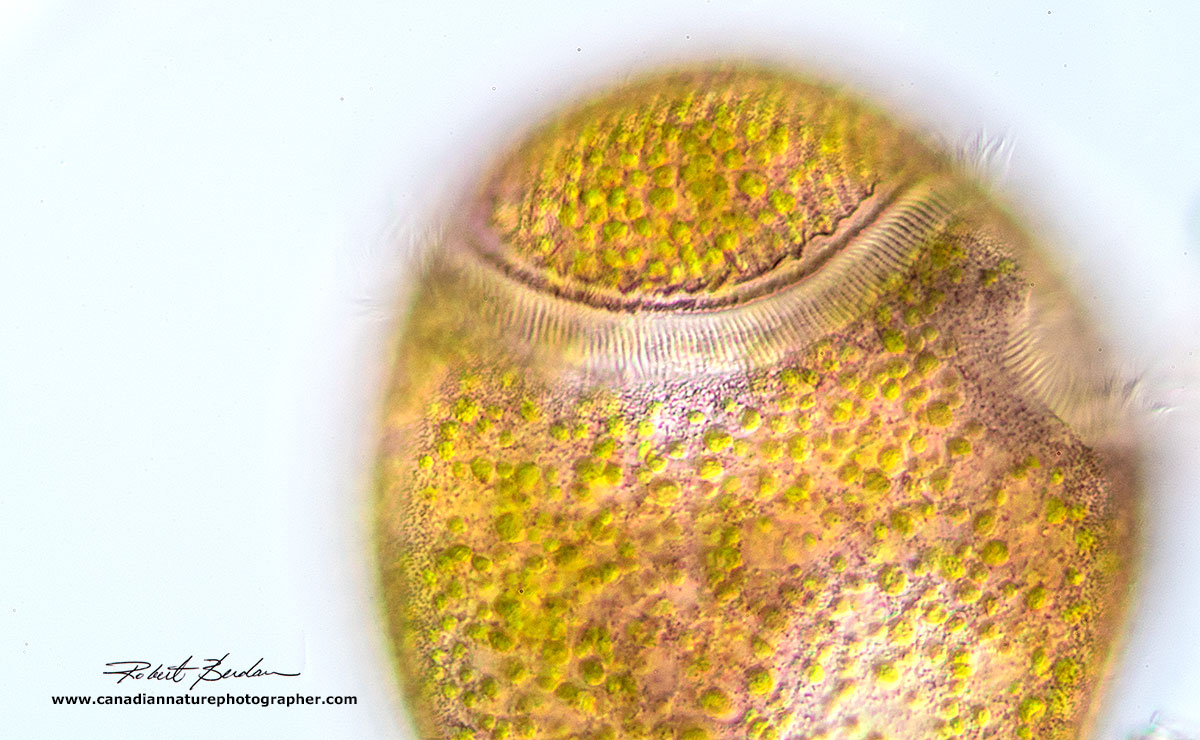
Above 2 photos show part of the Stentor amethystinus in bright field microscopy showing the green endosymbionts, the red-purple cortical granules and the larger membranelle band of cilia. 200X. Stentor amethystinus.

Stentor amethystinus - purplish red cortical granules with 1-5 ellipsoid macronuclear beads
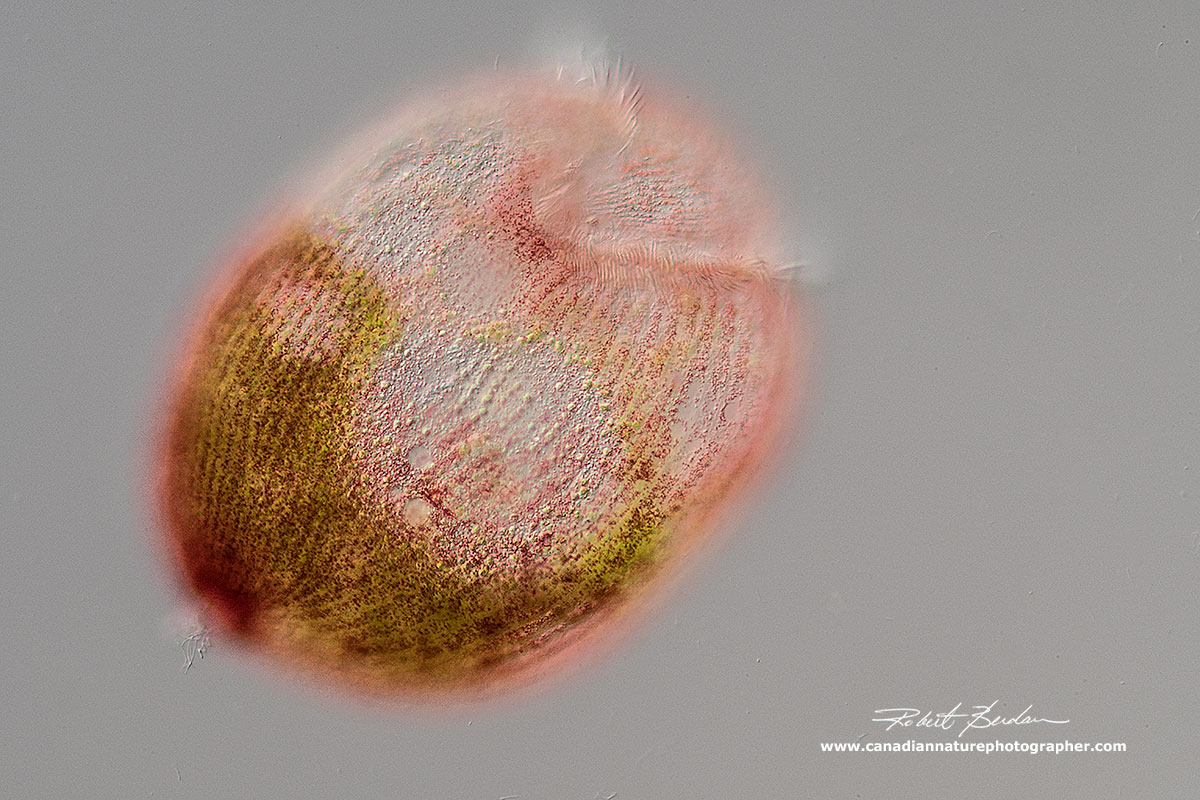
Stentor amethystinus - purplish red cortical granules with 1-5 ellipsoid macronuclear beads
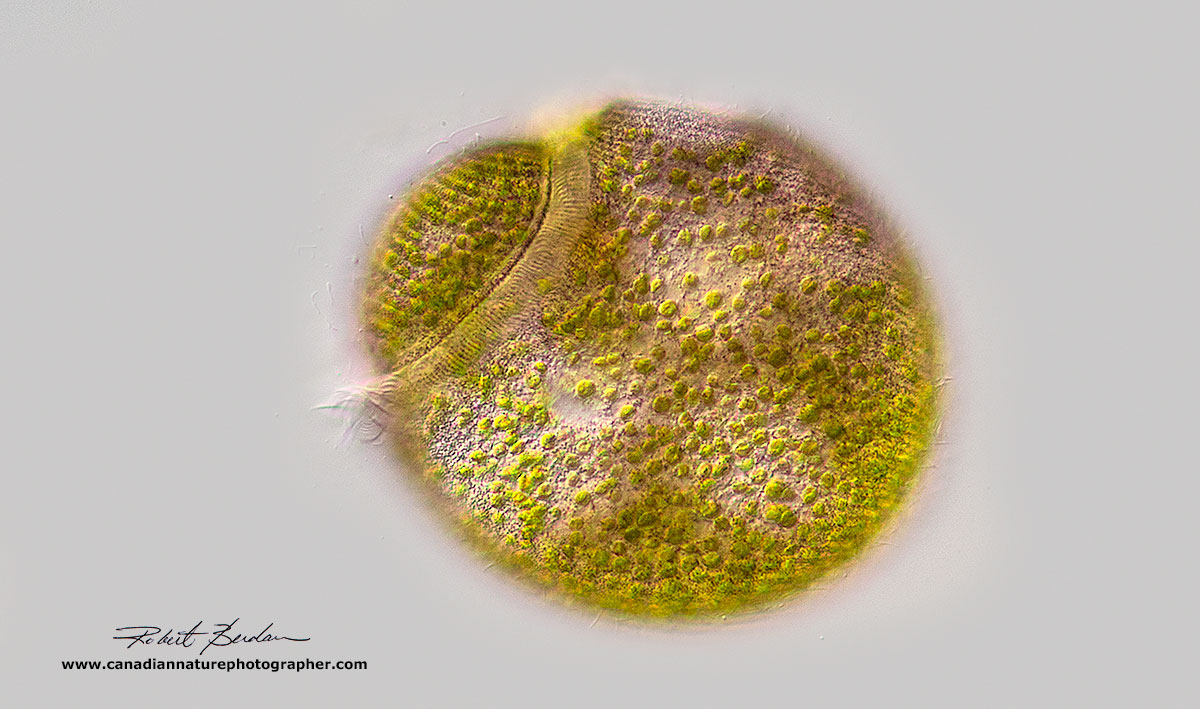
Above three photos show Stentors with endosymbiotic algae and the variety of shapes it can take. The number of symbionts can vary, but usually there are usually several hundred. DIC microscopy 100X Stentor amethystinus.
Stentors are of interest for several reasons. First their large size makes them conspicuous in pond water and when the bloom they can clog water filters. Their large size has also made them useful for some scientific studies. Scientists have shown that these single celled animals are capable of simple forms of learning - the fact is they can do so in the absence of a nervous system is fascinating. These animals have also been used in regeneration experiments and it has been shown that a piece of the Stentor if it includes part of the macronucleus can regenerate an entirely new animal.

Stentor sp showing the large membranelles on the anterior end 100X DIC microscopy
Most or perhaps all Stentors have sensory cilia and they have the ability to build a lorica (vase like attachment) that may protect them and is formed under optimal conditions when they attach to the substratum. I have never seen a Stentor with a lorica. Some of Stentors are able to create resting cysts so that when the pond dries up they can survive until water returns. Many pond organisms are capable of doing this as it essential to their survival. In activated sludge used in wastewater treatment the presence of Stentors indicates good operating conditions.

Stentor sp with few beads macronucleus and endosymbiotic green algae 200X DIC microscopy

Above Stentor amethystinus with endosymbiotic algae and red-purple cortical granules 200X DIC microscopy.
Stentors feed on many kinds of food including other ciliates, diatoms, small rotifers, worms, flagellates, bacteria and some are cannibals. They are efficient filter feeders. They exhibit both asexual and sexual reproduction with a generation time of about 3 days.
Stentors in Research
RNA splicing and structural inheritance were first discovered in ciliates, however some studies in ciliates can not be generalized to other animals, and some ciliates even have different genetic sequence (codons) coding for some amino acids. RNA interference studies could be performed by feeding Stentors with bacteria expressing long double-stranded RNA corresponding to a gene of interest for several days. This would knock out the gene products and one could observe the effects on these organisms.
Stentor has also been used in microsurgical experiments showing structural inheritance i.e. that some supramolecular organization of the pellicle can be inherited directly without DNA from the nucleus (see reference below to V. Tartar who developed many of the microsurgical techniques and studied regeneration). Almost any piece of Stentor can regenerate a new cell if the piece contains part of the macronucleus and a small portion of the orginal membrane\cortex. Stentor may be useful for studying wound healing at the single cell level and pattern formation.
Stentor could also be a useful model for studying simple forms of learning. Work from David Wood has shown the Stentor can habituate to mechanical stimuli and remain habituated for hours (i.e. with continued stimulation it responds less to the stimulus).
To find Stentors head to your local pond with some jars. To see them you can use a 10X magnifying loupe but the best way to see them is using a light microscope. I find it helps to take water from my pond collecting jars and spin the water samples in a clinical centrifuge (purchased on Kijjii) for a few minutes - this concentrates the larger organisms and Stentors in the bottom of the tube - I discard about 90% of the water and only use the concentrate on the bottom. You can purchase hand crank centrifuge for $165 on Amazon.ca or on Ebay.
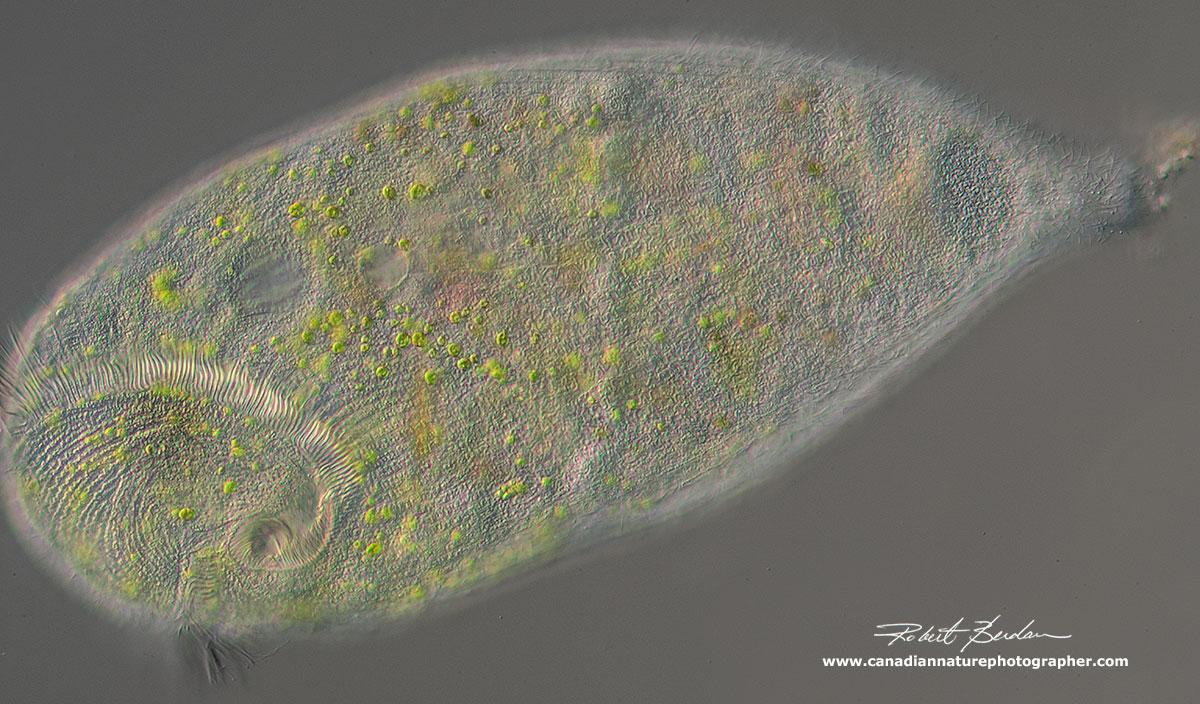
Stentor sp with endosymbiotic algae - DIC microscopy 100X
Notes on Photomicrography
To photograph the stentors I attached my Nikon D500 and D800 cameras to my microscopes: Zeiss Axioscope with DIC, Olympus E microscope with Phase contrast and Nikon Optiphot with Polarizing optics. I attached the cameras to my laptop via a USB cord and used free software Digicam control to trigger the cameras and grab the images. I used ISO speed of 200-1600 and edited the images in Adobe Photoshop. See my previous photomicrography articles and Tips on Photomicrography to learn more about how I take photomicrographs. To slow the Stentors down I compress the coverslip by drawing water out from below the coverslip using a piece of paper towel. Vince Tartar uses methylcellulose to slow ciliates down and I will trying this technique shortly.
I welcome any comments, questions or image purchase enquiries by email - rberdan@scienceandart.org

Stentor amethystinus viewed by DIC microscopy 100X
References & Links
V. Tartar (1961) The Biology of Stentor. Ulan Press - PDF available for purchase or View online for free at Internet Archive.
E. Newman (1972) Contraction in Stentor coeruleus: A Cinematic Analysis. Science 47: 447-449.
JT Randall and SF Jackson (1958) Fine structure and function of Stentor polymorphous. J. Biophys. Biochem. Cytol 4:807-830 - Download Free PDF
B. Huang and D. R. Pitelka (1973) The Contractile Process in The Ciliate, Stentor coeruleus. J. Cell Biology: 57, 704-728. PDF (B. Huang was the wife of my Ph.D. supervisor Dr. Norton Gilula, this is primarily an electron microscopic study of Stentor coeruleus).
W. Foissner (2002) Stentor in Encyclopedia of Life Scienes - download PDF
W. Foissner and S. Wölfl (1994) Revision of the genus Stentor Oken (Protozoa, Ciliophora) and description of S. araucanus nov. spec. from South American lakes. J. Plankton Research. 16: 255-289. Contains a revised biological key to the Stentors - download PDF
G. Hölfe, S. Reinecke, U. Laude and D. Spitzner (2014) Amethystin, the Coloring Principle of Stentor amethystinus. J. Natural Products. American Chemical Society - download PDF.
B. Taylor - web site "It came from the Pond" all about ciliates, the web site author has helped me identify and learn more about many ciliate species including Stentors.
Photomicrography and Video of Protozoa, Volvox and Rotifers - includes a movie of Stentors by R. Berdan article on this web site.
Pennard Labs web site - learn more about protozoa and see amazing photographs, these guys have a scanning electron microscope in their home lab - I am envious!
Infusoria - web site showing early paintings of Stentor species - www.infusoria.org.
Microsurgery Experiments on Stentor by V. Tartar - Chapt 9 PDF
Stentor coeruleus - Curr Biol (2014) Vol 8: 783-784 overview - download PDF
Stentor Genome Database -stentor.ciliate.org
Buy live Stentors from Boreal (Ward) Science - they will send you a tube of them for $11.60. I purchased a tube in February after buying my new microscope as I could not wait for the ice to melt off the ponds! All the Stentors photographed in this article except one which is noted were free living animals collected in local ponds. Note magnifications indicated are approx.
Authors Biography & Contact Information

Robert Berdan is a professional nature photographer living in Calgary, AB specializing in nature, wildlife and science photography. Robert retired from Cell\Neurobiology research to take up photography full time years ago. Robert offers photo guiding and private instruction in all aspects of nature photography and Adobe Photoshop training - including photomicrography, macrophotography.
Related Microscopy Articles by Robert Berdan on this web site
1. How to Collect and Photograph Water Bears (Tardigrades).
2. Tips on How to take Better pictures with a Microscope
3. Microscopic Pond Organisms from Silver Springs Calgary
4. Microscopic Life in Ponds and Rainwater - Pond Scum I
5. Photographing Microscopic Plant and Animal Life - Pond Scum II
6. Photomicrography and Video of Protozoa, Volvox and Rotifers
7. Home Microscopy Laboratory for Photomicrography
8. The Art & Science of Photomicrography with Polarized Light
9. Photographing Through a Microscope Photomicrography - Inner Space
10. Focus Stacking comparing Photoshop, Helicon Focus and Zerene
11. Rheinberg Filters for Photomicrography
12. Scanning Electron Microscopy - Photography
13. Photomicrographs of Diatoms from 1877 by John T. Redmayne
Email at: rberdan@scienceandart.org
Web site: www.canadiannaturephotographer.com
Phone: MST 9am -7 pm (403) 247-2457.
Click on the buttons below and share this site with your friends