
Photographing Ciliates
by Dr. Robert Berdan
July 17, 2018

Spirostomum minus (top) and Urocentrum turbo (bottom) - both are common ciliates in fresh water. DIC (differential interference microscopy) A micron = 1\1000 of a millimetre.
Introduction to the ciliates
You may have been introduced to Paramecium in biology class, a single celled (unicellular) "simple" ciliate found in pond water. Only ciliates are not simple but some of the most complex cells on the planet. They have an intricate cytoskeleton (support network of microtubules and filamentous fibers), cilia of several different sorts, 2 different types of nuclei and come in a wide variety of shapes and sizes. Ciliates can be found anywhere there is water; in ponds, streams, lakes, oceans, soil and the water in and around plants, mosses and liverworts. It isn't known how many species there are but about 9000 species have been described and estimates suggest there may be as many as 30,000 or more. Most ciliates are microscopic in size, but a few get as large as 4 mm in length (e.g. Stentor).
Cilia are small hair-like structures that cover most ciliates during some stage of their life cycle. Cilia means "eye lash". The organisms may have different sized cilia. Cilia are used for locomotion and also to direct food into their mouth. Cilia may be fused into long sheets (undulating membranes), or arranged as a series of short pennants; they may form movable tufts (cirri), or stiff bristles (setae). However, it's not the presence of cilia that characterize this group, but the presence of two types of nuclei - a macronuculeus and micronucleus.
Spirostomum minus - DIC microscopy
The macronucleus is often large and sometimes is easily seen inside the cells, and it is also polyploid (contains multiple copies of the genes). The function of the macronucleus is to regulate metabolism and day to day functions of the cells. There may be one or more micronuclei that are involved in genetics and exchange of DNA.
Reproduction usually occurs by binary fission across the long axis and they can also undergo conjugation where they exchange genetic material with another ciliate of the same species. Conjugation involves partial fusion of paired cells allowing mutual exchange of genetic material. In syngamy the two cells fuse completely. They can also reproduce using a variety of other methods (see below). After about 200 cell divisions some ciliates show signs of aging and the reorganization of their DNA during conjugation somehow reestablishes their vitality and makes them young again.

Most ciliates divide by binary fission, but they may multiply using a variety of other methods shown above. Diagram courtesy Wikipedia by Deuterostome.
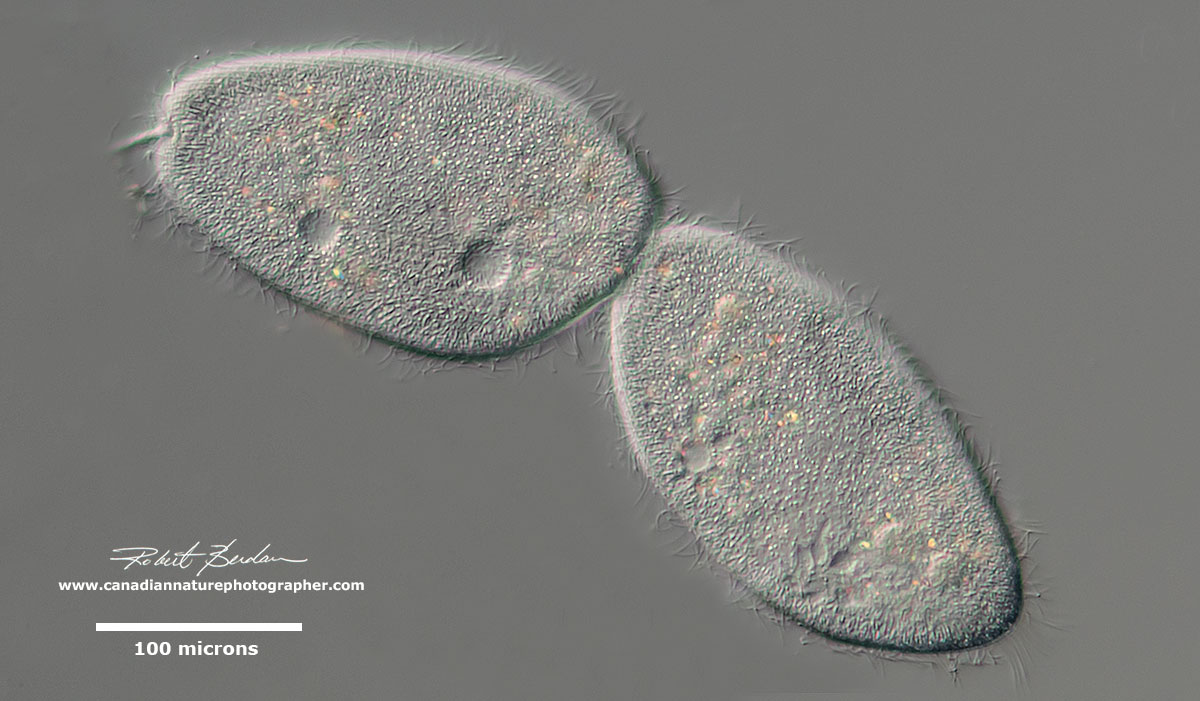
Paramecium caudatum dividing. I usually see ciliates dividing in the evenings. They usually continue to move about during division perhaps to avoid predation.
Ciliate dividing

Two scuticociliates Dexiotricha granulosa with ring shaped glycogen granules in the cytoplasm joined together possibly undergoing conjugation. (ID courtesy of Bruce Taylor - YouTube Video).

Two more ciliates that appear to be undergoing conjugation.

Palintomy a process during which a large parental cell undergoes a rapid sequence of repeated divisions without intervening growth. Arrows point to the boundaries between 4 Colpoda ciliates. Several frames were extracted from a movie and focus stacked, DIC microscopy 600X.
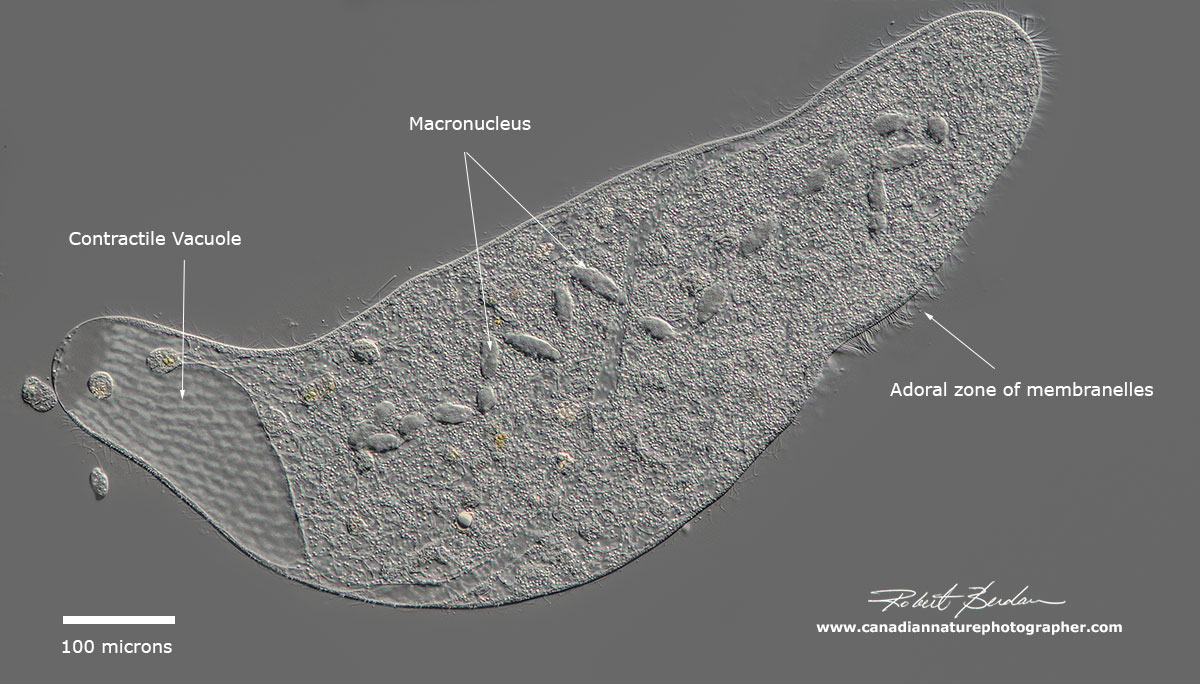
Most ciliates ingest food through a cytosome, once ingested food is enclosed in a food vacuole. The digested food contents are released by the ciliates anus which may be well defined (cytoproct). Ciliates also contain one or more contractile vacuoles which functions to remove excess water or ions that flow into the cells by osmosis. In Paramecium you can watch the contractile vacuole expand and contract at regular intervals. The position of the contractile vacuole can be useful in identifying some species.

Spirostomum minus with adoral zone of membranelles (larger cilia) on the right side of the body extends almost half the body length. 600X DIC microscopy.

Above, Spirostomum minus posterior end, the contractile vacuole is in the darker region and you can see a spiral pattern on the pellicle made up of cortical granules. 600X DIC microscopy.
The ciliate cortex (outer covering) exhibits features used to characterize and identify different species along with their shape and approximate size. The cortex is often divided into two regions the somatic (body) and the oral regions. The ciliary structure around the oral regions is important in classifying the organisms. An element of the cortex is the Kinetid composed of one or more kinetosomes with their fibrils and cilia. Kineties are simply ciliary rows (kinety is a single row of cilia). Somatic kinetids may have 1, 2 or more Kinetosomes through which the cilia are formed. The presence and distribution of kinetids on the body surface is a characteristic of the taxon.

Streamlined like an alien spaceship this ciliate is able to swim quickly. I found it in moss while searching for water bears (Tardigrades). This ciliate was flattened dorsal - ventrally. Phacodinium metchnikoffi is apparently a rare ciliate (see M. Kruetz and W. Foissner publication page 141 - free PDF with excellent photomicrographs).
Some ciliates lack a cystosome and acquire nutrients at the cell surface by adsorption and pinocytosis (ingestion of small membrane vesicles). Suctorians are a group of ciliates that are sedentary as adults without cilia and ingest food on the tips of long tentacles (see below).
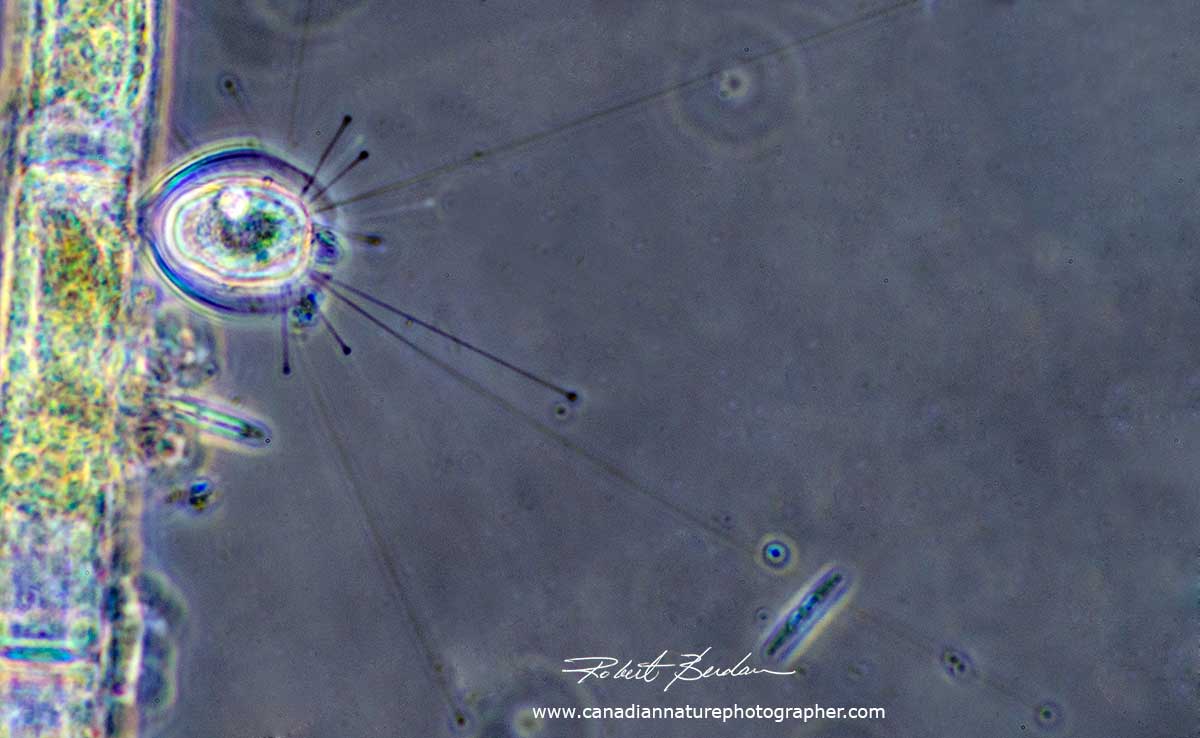
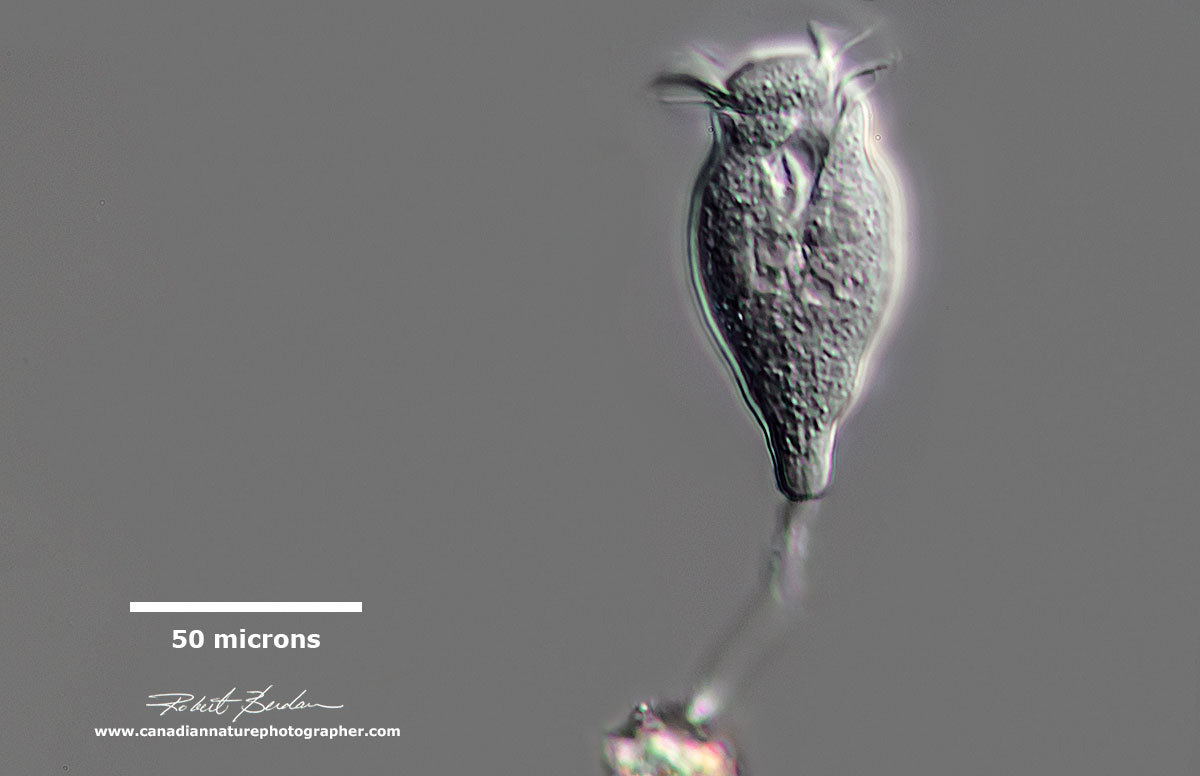
Above - a Suctorian with long tentacles used to capture prey (Podophyra fixa). Phase Contrast microscopy, Olympus E microscope. These ciliates only have cilia during their early stage. They often reproduce by external budding.

Spathidium spathula found in moss while searching for water bears, the oral region (top) is conspicuously swollen.
Cilia are not restricted to ciliates, but are found associated with many other animals and in fact cilia are responsible for removing dust and other pathogens from our own lungs. However, in many ciliates, cilia allows them to move about quickly, back up and even spin making photography of these organisms challenging at times. To get sharp pictures I need to use fast shutter speed (1\500 of sec or faster) or trap the animals by removing water from below the overlying coverslip with filter paper (see my article Tips on How to Take Better Pictures with a Microscope). The compression technique seems to work with some of the larger ciliates. Another way to slow ciliates down is to increase the viscosity of the water (e.g. adding methyl cellulose). Gently heating the microscope slide for a few seconds under a flame may also stop or slow them down but often it cooks the organisms and they burst shortly after. It is also possible to use a high speed flash to stop the motion of some ciliates.

Urocentrum turbo (Family Parameciidae) - a fast moving ciliate found in pond water often in association with Paramecium. They spin and move erratically.
The fastest Ciliate on the Planet?

Halteria sp - side view showing some of the long bristle like cirri

Halteria sp - dorsal view looking down showing the adoral zone of membranelles. This organism can move hundreds of times its body length in less then a second and often disappears out of the field of view when observing with a light microscope.

Diagram of Halteria from C. R. Curds (1982) British and Other Freshwater Ciliated Protozoa. Cambridge University Press. London (page 439).
Halteria is a small but common ciliate found in pond water. What makes this ciliate interesting is that one moment it will be lying still and then appear to jump out of the field or across the slide at very high speed - like jumping to warp drive. A study using high speed digital video recorded the ciliary beating of Halteria grandinell to be between 105 and 260 Hz (cycles per second). These frequencies are the highest observed for cilia and flagella reported to date (2005).


Above pictures show the giant ciliate Spirostomum minus and smaller Paramecium caudatum.
Identifying different ciliates is challenging because there are so many different shapes and varieties and many of them can move quickly. The taxonomy of these organisms is also changing as more researchers apply molecular biology techniques to analyze similarities and differences.
In this article I show some ciliates that are relatively common. I almost always encounter new ones that I have never seen before in any jar of pond water I collect. The great variety of ciliates intrigues me. I have purchased several books and and guides to help me identify many of the organisms and I also send pictures to a friend and ciliate expert (Bruce Taylor) for verification or correction. However, one doesn't have to be able to identify each species in order to appreciate them.
Ciliates can be found in bodies of water all year long even under the ice. One can also culture ciliates by producing a hayinfusion where one puts hay in water with a small amount of powdered milk, wheat or rice grains (read more on the Microbe hunter). The ciliates feed on the bacteria that in turn feed on the powdered milk or grains. I made several hayinfusions when I was younger when I wanted to view protozoa throughout the winter months. You can also purchase ciliates for classroom or private study e.g. Boreal Science. I purchased some Paramecium and several other invertebrates from Boreal this past February - a small tube is only $12.00. The rest of the year any body of water is likely to have some ciliates in it. You can even collect rain water from your roof to see some ciliates and other protists (see my article Microscopic Life in Ponds and Rainwater). I also use a centrifuge that I purchased on Kijjii to concentrate some of the water samples. Generally I spin the water samples for 2 minutes at 1800g - though often I can find plenty of ciliates and other organisms by sampling the water from a jar and\or including some water plants.
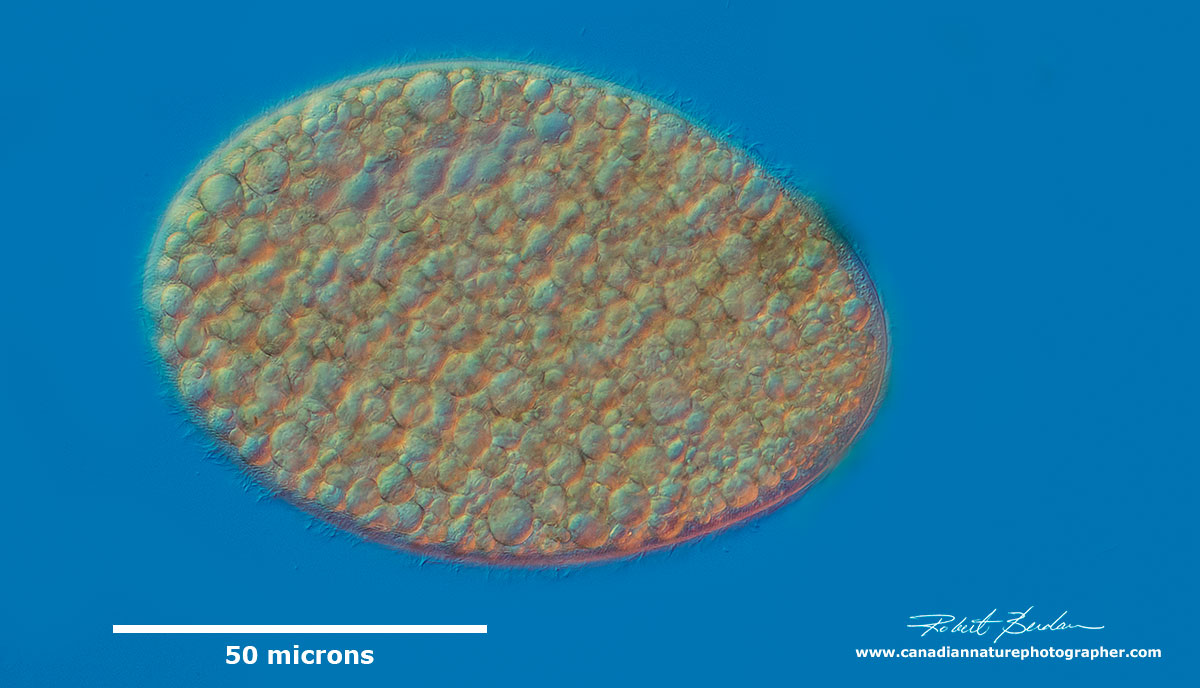
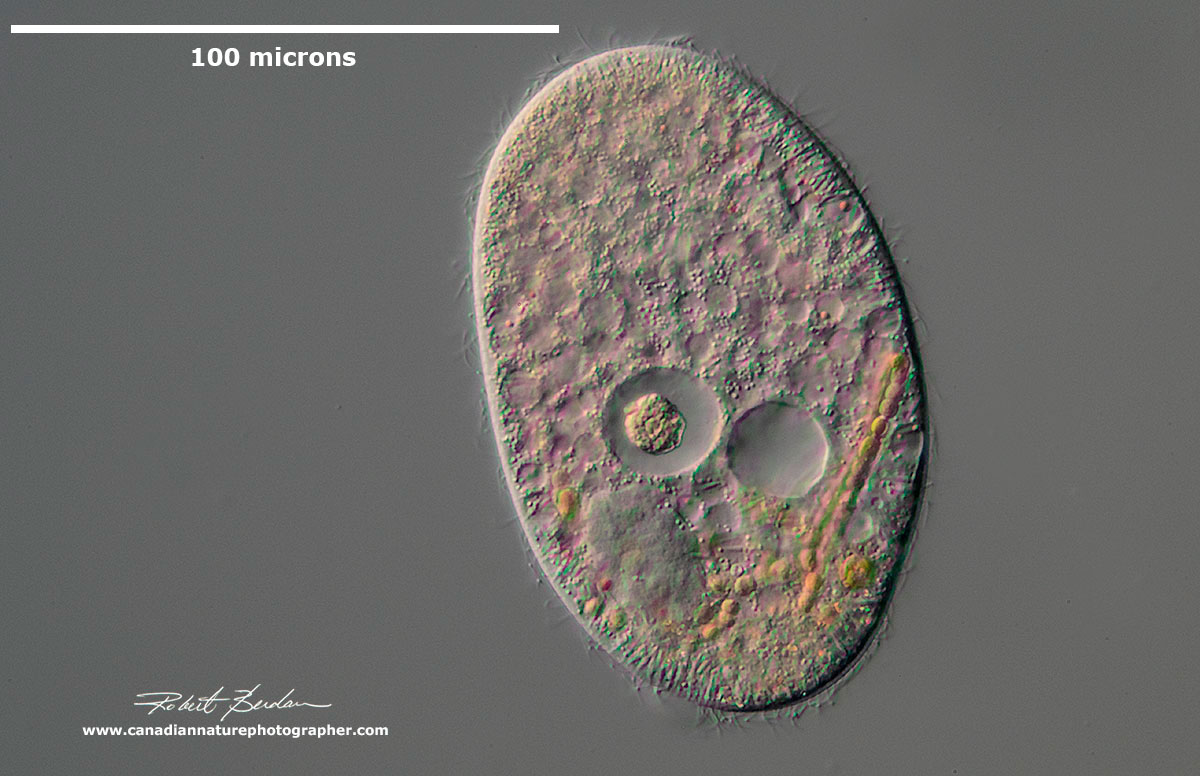
Colpoda cucullus (Class: Colpodea). Ciliate which in well-nourished specimens is crammed with up to 200 compact, 5-8 microns sized food vacuoles. These medium sized ciliates are common and often bean shaped. They are one of the earliest ciliates to appear in a hay infusion. They are frequently found in soil and moss because of their ability to form cysts. They feed primarily on bacteria. Learn more and visit EOL web site for a more detailed description of this species.

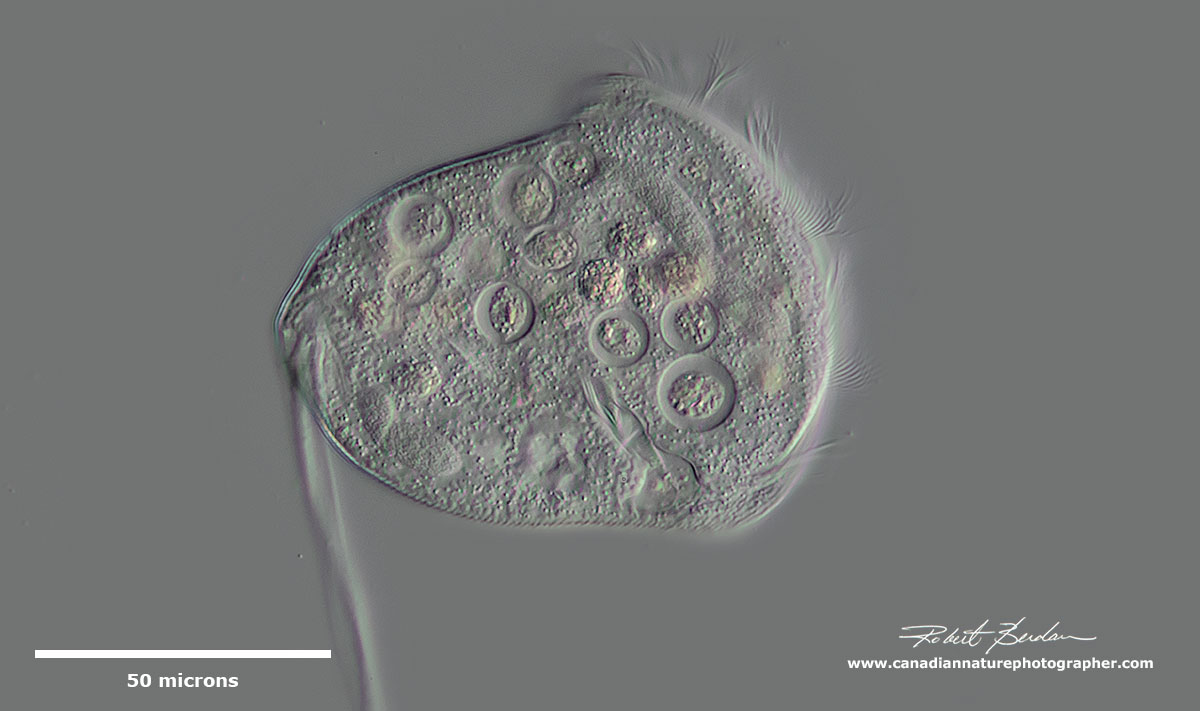
Unidentified medium sized ciliate with membranelles around the anterior end, several large macronuclei and conspicuous contractile vacuole. Heterotrich.
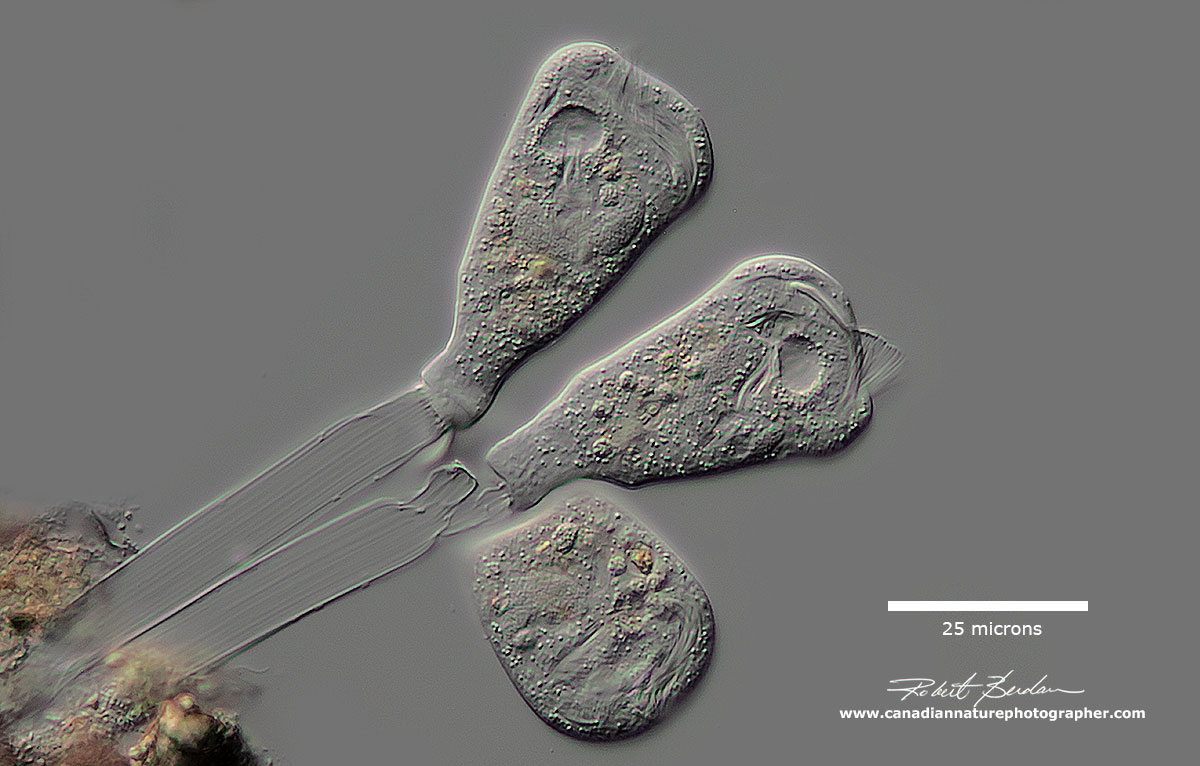
Vorticella or Pseudovorticella is a Peritrich ciliate which attaches to the substratum via a retractable stalk. The stalk can contract quickly and the ciliates are often found in groups attached to underwater plants. The cilia sweep bacteria into their cytosome along with other small organisms like algae - essentially they are filter feeders. The have a large C shaped macronucleus (see more pictures in my article on Protozoa). DIC microscopy - learn more about Vorticella. Note Pseudovorticella has a sort of brickwork pattern, whereas Vorticella has lateral corrugations on its surface.
Peritrich in which the stalk was short and did not contract like those in Vorticella. Class: Oligohymenophorea; Subclass: Peritrichia - epistylid.
Peritrich - Epistylis sp on broad flat stalk that is not contractile.

Above two photos - These are the swarmer (telotroch) phase of a peritrich ciliate - probably Vorticella. When swarming, they shed their stalks, become somewhat cylindrical and develop a wreath of cilia at the posterior. Most peritrichs have a telotroch phase (identified by Bruce Taylor). See video by Bruce Taylor and read additional information about telotrochs on www.microscopy-uk.org.uk.

At first I thought this was Stylonchia mytilus a very common ciliate in pond water and soil. It is has three long cirri at the posterior, and cilia grouped into membranelles alongside the mouth and cirri over the body. However ciliate expert Bruce Taylor suggested that this is probably Tetmemena as three members of Stylonchia were moved to this genus several decades ago.
The easiest way to observe ciliates and other protozoa is to collect water from a pond, ditch, stream edge, lake, estuary, or pour some bottled water or distilled water over some dry moss or soil in a dish and wait an hour then examine some of the water on a microscope slide with a microscope. A simple microscope with brightfield illumination will work fine. I started with a $30 toy microscope. An interest in microscopy lead me into a career in cell biology and nature photography. Microscopes with Phase contrast or Differential Interference will make it easier to see the ciliates as most of the protists are translucent, but any decent microscope with 100-400X magnification will permit you to see and study these organisms. You can also stain the ciliates though this usually kills them and you don't get to observe their interesting behaviour - for more information on specimen preparation see the free PDF - Protocols in Protozoology.

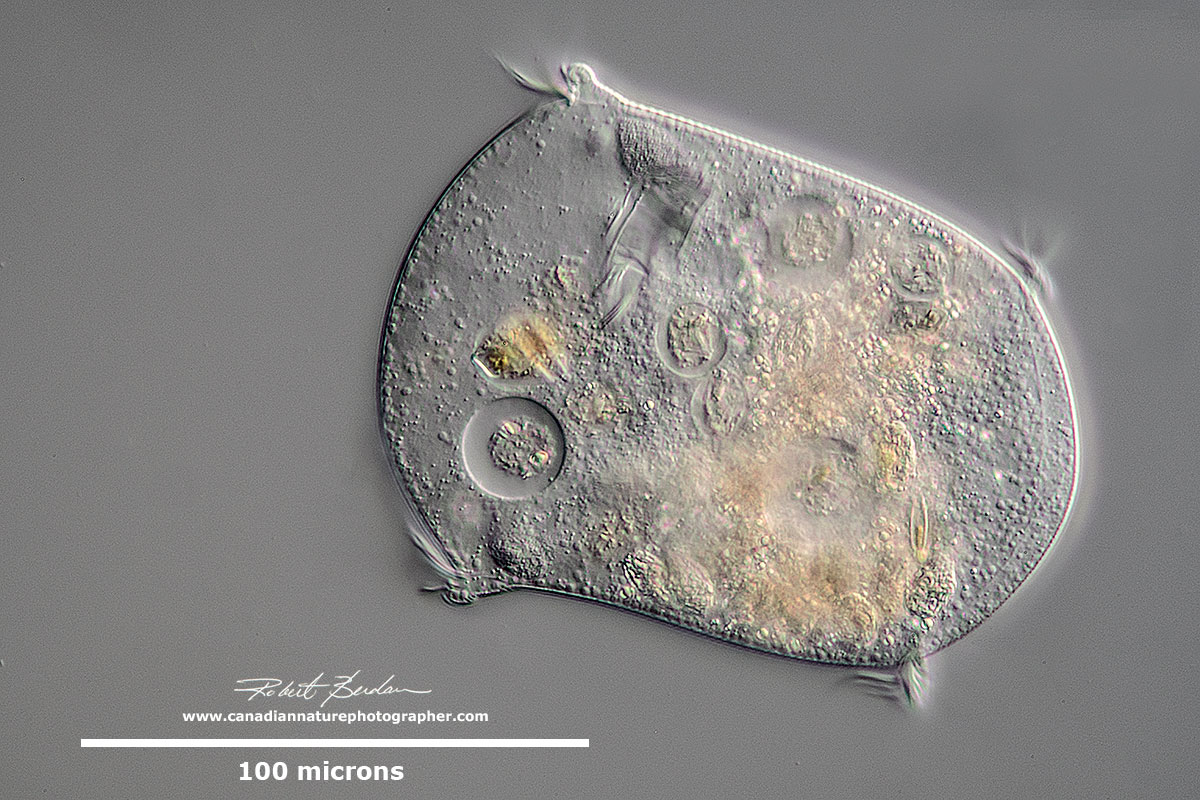
Hypotrich - unidentified ciliate. Hypotrichs possess compound ciliary organelles called "cirri," which are made up of thick tufts of cilia, sparsely distributed on the ventral surface of the cell.
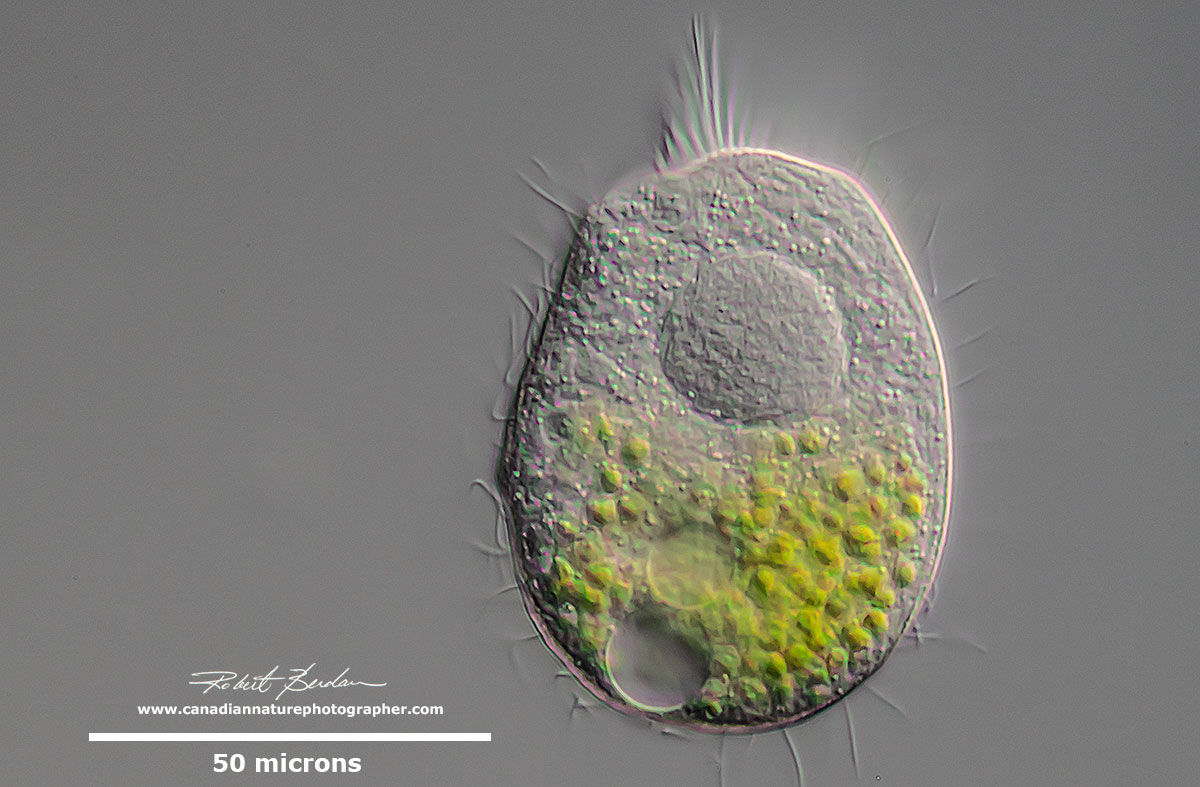

Above two photos of unidentified ciliate with endosymbiotic algae and single large Macronucleus. Some ciliates ingest green algae (Chlorella sp) or acquire them from previous generations. These algae are not digested but form a symbiotic relationship with their host. The algae share sugars they produce via photosynthesis with their host and the host in turn provides them with some protection and nutrients. It is not fully understood why some ingested algae are not digested and how the algae are able to communicate with their host to release sugars to them. Symbiotic algae may be passed down through the generations, and can be closely integrated with the host cell and may even be retained in cyst form. In laboratory conditions, some ciliates can be stripped of their symbionts and reinfected with new ones, but it is not known how often this occurs in nature. Phylogenetic analysis shows that the strains of Chlorella in a variety of ciliates are related to one another and are distinct from "wild" chlorellae (Bruce Taylor personal communication - see this reference for additional information).

Stentor polymorphus with endosymbiotic algae inside. Bright field microscopy.

Ciliate filled with green algae (endosymbiotic) - possibly Holophyra ovum (suggested ID by Bruce Taylor).
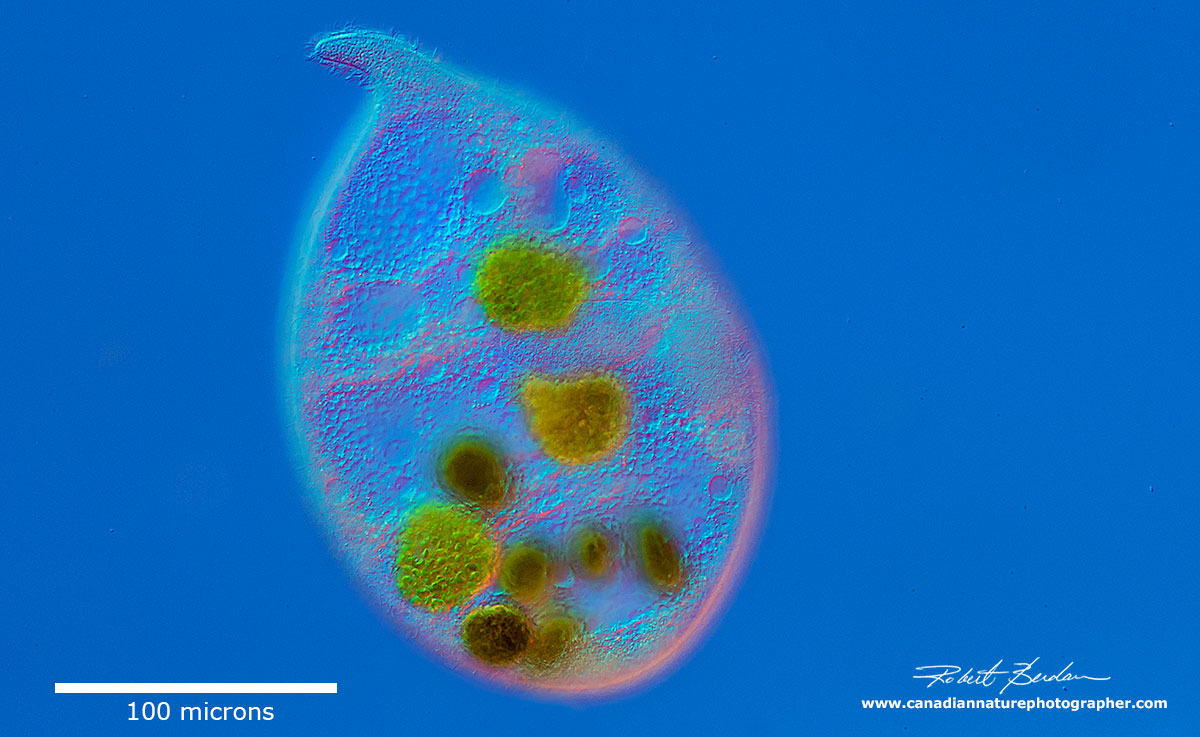
Trachelius ovum- this unusual ciliate would eject parts of its cellular contents when trapped by the coverslip in order to become a smaller organism. It's body appears to be very fragile and this may be a mechanism to avoid predators. Below you can the same organism after it shed its internal components. Mouth is round, located at the base of a short proboscis.
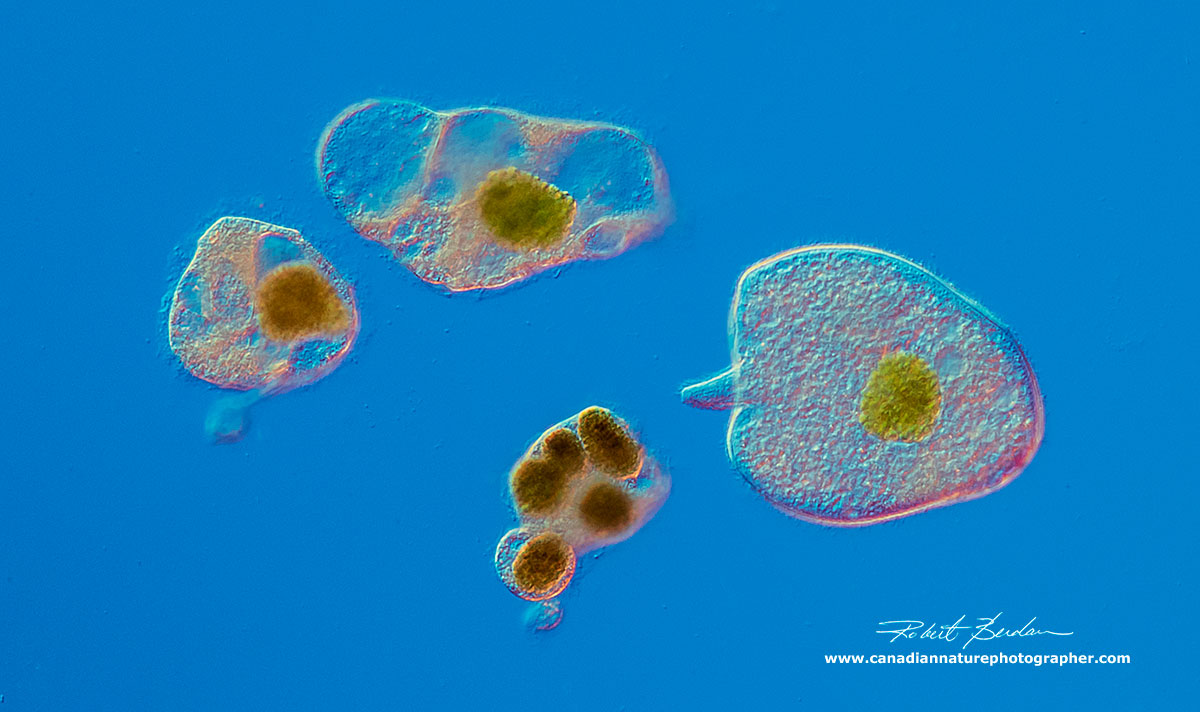
Trachelius ovum - next to the body parts it shed. This unusual behaviour has been observed before in Trachelius and reported in J. of Natural History by S. G. Foulke 2009 - describing observations by Mr. Charles Robertson in 1867. Also see observations by C. Gegenbaur (1857).

Trachelius ovum - after it expelled its body parts.

Flattened Trachelius ovum - sometimes under the pressure of the coverslip this organism would just explode into pieces.

Nassophorean Nassula ornata - its mouth leads to palisade of rods or nematodesmata seen inside the cell at the upper right (bright white area). Body ovoid and uniformly ciliated with a single macronucleus (white circle). They feed on blue-green algae and as the algae are broken down they can take on a variety of bright colours. Class: Nassophorea, Order Nassulida, Family: Nassulidae. Learn more and view videos of this microorganism.
Nassula ornata - ciliate with a single strand of blue-green algae (cyanobacteria - procaryote which are cells with no nuclear membrane) strand visible inside on the right (Anabaena sp).
Oral area seems to comprise more then 2\3 of the ventral surface. Lembadion lucens. Watch YouTube video of this ciliate.

Frontonia atra with dark pigment granules. I thought the dark granules might be bacteria living inside but they may simply be dark pigment granules. Ciliates in general have been found to host 60 different species of Bacteria.
You can stain ciliates with methylene blue, neutral red or Lugol's iodine, though this can or will kill them. Also you can use a technique called Darkfield microscopy which presents the ciliates against a black background (see above Nassula sp) and the organisms tend to light up white. This technique can be mimicked by putting a coin on the light source or under the condenser on a filter. Oblique illumination can be created by putting the condenser filter holder partially in front of the light source and this will creates a three dimensional like appearance of the specimens that resembles DIC microscopy. Of course you will need some glass microscope slides, coverslips and an eye dropper to put a drop of pond water on the slide for viewing.

Above one ciliate appears to be devouring another. I watched them for 45 min as they struggled often shaking violently as one tried to escape.
Cysts
Of the thousands of ciliate species known so far only one, Balantidium coli, is known to cause disease in humans. It's regular host is pigs and infection can occur by ingestion of water contaminated with cysts from this ciliate. Many freshwater ciliates, protozoans and other invertebrates are able to survive dessication by forming cysts. I see a lot of cysts in my pond samples but it's difficult to identify which group of organisms the cysts belong to except perhaps some of the cysts of rotifers were I can view their mastax (jaws) through the cyst enclosure.

Above is just one of hundreds of different unidentified cysts or eggs that I have seen in pond water. Cysts allow many microorganisms to survive dessication, but except for some rotifer and tardigrade cysts they are difficult to identify which organisms they belong to.

Lacrymaria olor a ciliate with long extensible "neck". This organism is sometimes referred to as "Tear of a swan". This ciliate usually has two macronuclei and one micronucleus. The body cilia are arranged in a spiral (see below). It has two contractile vacuoles one at each of its body and its neck can expand or shrink back into its body. (Class: Litostomatea, Family: Lacrymariidae).
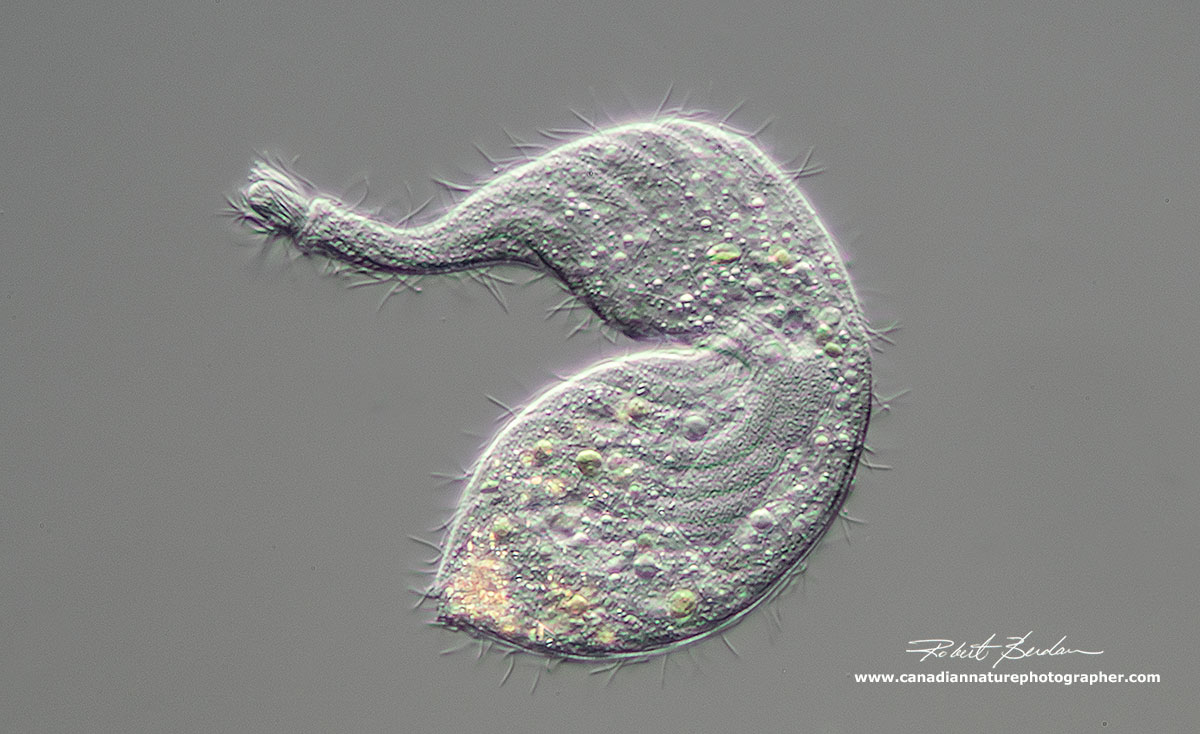
Lacrymaria olor contracted showing its flexibility and large oval macronucleus.

Lacrymaria olor showing macronucleus about 600X DIC microscopy

Paramecium caudatum DIC microscopy. I focused on the outer pellicle.
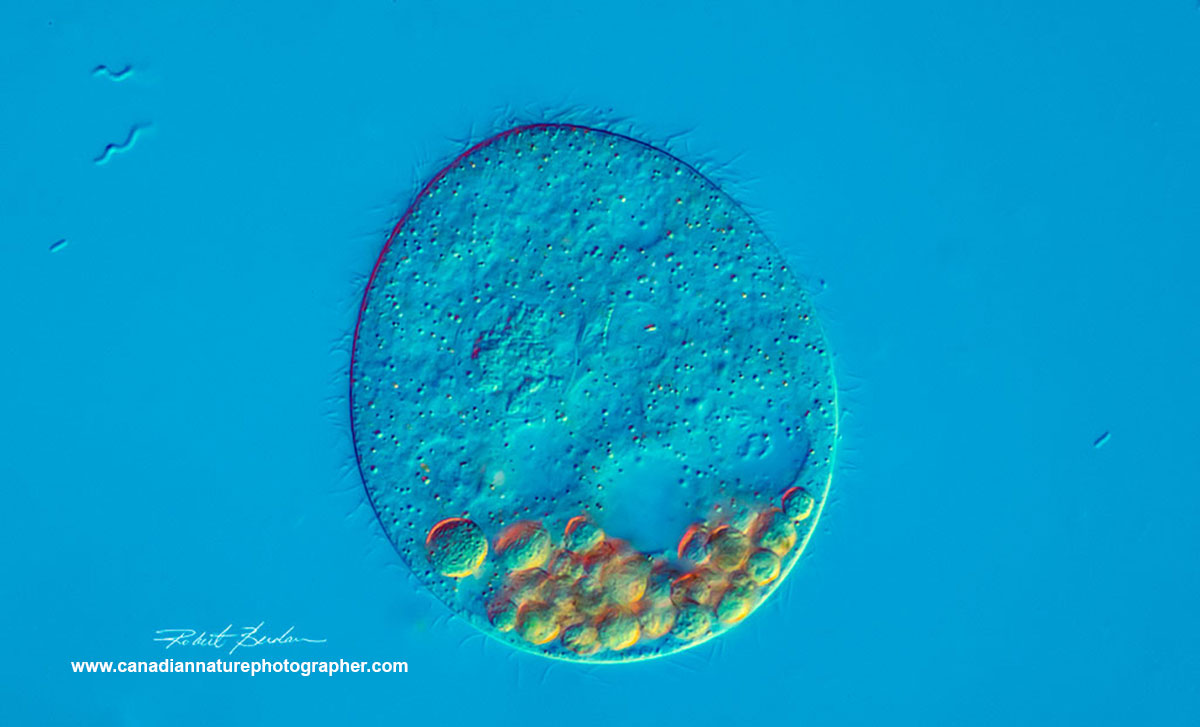
Unknown ciliate species? At the top left in the background of the picture are Spirillum "w-shaped" bacteria. The large coloured vacuoles in this ciliate have collected at the "bottom" of this specimen.

Loxophyllum helus is a common elongated ciliate with a neck region and rounded tail, two ovoid macronuclei (just visible inside near the center) and a single contractile vacuole (not visible). The ciliate can be up to 160 microns long.

Loxodes rostrum The cytoplasm in this ciliate is highly vacuolated and it appears to have "beak" shape at the anterior end. There is a small diatom that can also be seen inside near the center. Loxodes belongs to the group of Karyorelictea where the macronucleus does not divide. Loxodes in greek means "oblique, tilted". More photos and information about Loxodes on Pling factory.

Unidentified ciliate with moniliform macronucleus (beads on a string) and two contractile vacuoles (dark circles), no endosymbiotic algae - possibly a lisostomatean (e.g. haptorid).

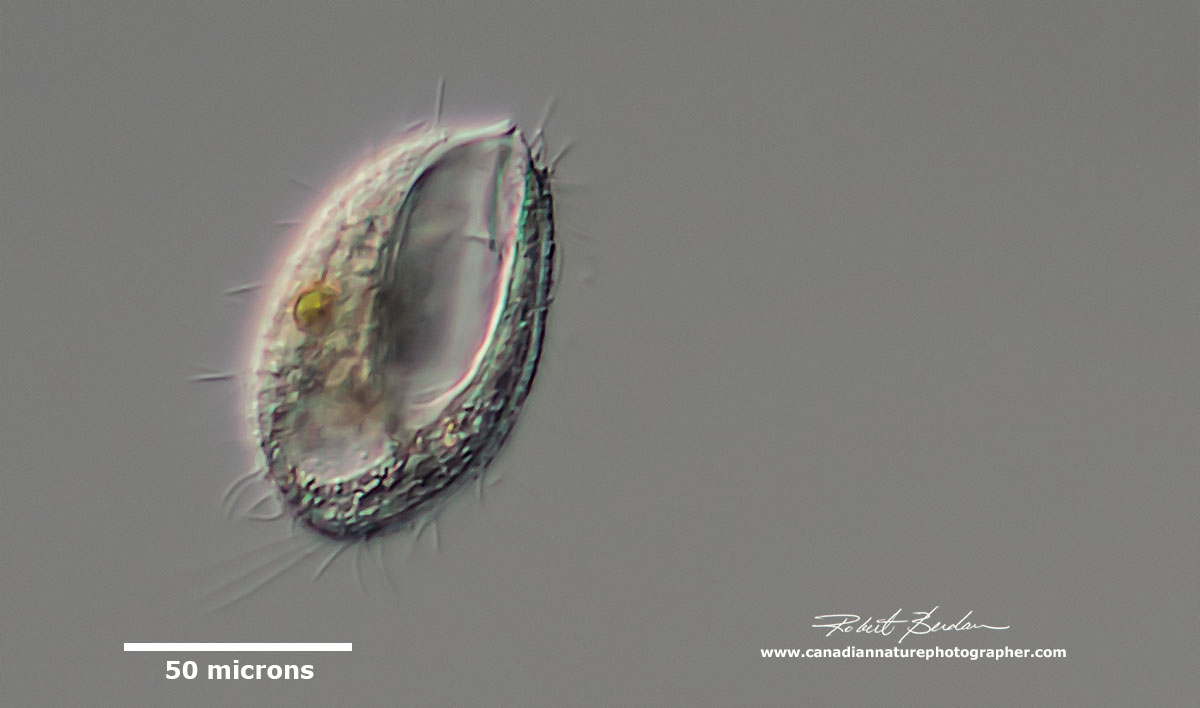
Euplotes sp?

Ciliate - spindle shaped hypotrich possibly contracted Stichotricha or Stongylidium (ID suggested by Bruce Taylor)

Ciliate with an adoral zone of membranelles, it appears to be slightly flattened by the coverslip.

Coleps hirtus has an elaborate ornamentation of the ectoplasmic plates which make up its test (shell). These plates are located inside alveolar vesicles of the cell cortex and contain calcium carbonate plates. DIC microscopy read more.

Coleps hirtus Darkfield microscopy. Coleps feeds on bacteria, algae and flagellates. It uses toxicysts, which are organelles which contain a toxin to stun or kill its prey.
Classification of ciliates (according to Wikipedia)
Domain: Eukaryote (cells with a nuclear membrane) - others include: Procaryotes, Archaea, - Viruses and Prions are currently being considered.
Kingdom: Chromista
Phylum: Ciliophora - protozoans with cilia, 2 types of nuclei (>3500 species)
Subphylum: Postciliodesmataphora - share stacks of postciliary microtubular ribbons associated with somatic kinetosomes, and called postciliodesmata
Class: Heterotrichaea - adoral zone of membranelles macro nuclei divide via extra macronuclear microtubules. e.g. Stentor
Class: Karyorelictea - most are marine except Loxodes, alveoli absent in most
Subphylum: Intramacronucleata - dividing macronucleus that relies on intramacronuclear microtubules
Class: Armophorea - small, aneaerobic ciliates - e.g. see EOL website
Class: Listomatea - e.g. Lacrymaria olor and human parasite Balantidium coli
Class: Colpodea - e.g. Colpod sp. small bean shaped ciliate with uniform cilia
Class: Nassophorea - mouth leading to palisade of rods or nematodesmata
Class: Phyllopharyngea - in some only young have cilia e.g. chonotrics and suctoria
Class: Prostomatea - e.g. Coleps - charateristics see EOL website
Class: Plagiopylea - ciliates common in anaerobic habitats
Class: Olighymenophorea - ventral groove leads to cytosome e.g. Paramecium
Class: Protocurziea - see examples - ancestral form of ciliate?
Class: Spirotreachea - - prominent oral cilia e.g. Euplotes
Class: Cariotrichea - only one species Cariacothrix caudata from Venezuela
Subphylum: Mesodiniea - new group of cliates - more information (for Geeks only).

Spirostomum minus in a contracted state. The contractile vacuole can be seen on the left in the posterior end. The zone of membranelles on the top right takes up about half the body length.
Photomicrography
For details on how I took these pictures please see my article - Tips on How to Take Better Pictures with a Microscope - Photomicrography. I used a Nikon D500 and Nikon D800 cameras, Zeiss Axioscope with DIC, Bright field, and Darkfield lighting, and controlled my cameras with free software Digicam control. Images were captured onto an Alienware laptop and processed in Adobe Photoshop. In many instances I needed to stack 2-12 images to increase the depth of field - for more information on this procedure see my article on focus stacking.
Summary
Ciliates are among the most complex and diverse unicellular organisms on the planet that form an important component of aquatic ecosystems acting as predators and providing nutrition for organisms at higher trophic levels. Ciliates are also important in the treatment of sewage water and water purification (Madoni 2011). My goal in this article was to share a small selection of photomicrographs and some interesting bits of information about these organisms and hope that it might inspire you to learn more about them.
Online References most or all the references can be found by a Google search
(2016) F. Gao et al. The All-Data-Based Evolutionary Hypothesis of ciliated Protists with a Revised Classification of the Phylum Ciliophora (Eukaryota, Alveolata). Nature scientific reports
(2016) First Record of Two Spirostomum Species of ciliates from Korea - download PDF.
(2016) T. Pattinson. The Freswater Microscopist - Part 2. Available on Blurb.com
(2015) T. Pattinson The Freshwater Microscopist. Available on Blurb.com
(2014) S. I. Fokin & V. Sera. The Hidden Biodiversity of ciliate-Endosymbionts Systems. Mini Review SciMedCentral - download PDF.
(2013) Morphology and 18S rDNA gene sequence of Spirostomum minus and Spirostomum terres from Rio de Janeiro, Brazil - Zoologia 30: 72-79 - download PDF.
(2012) C. Shao et. al. A redescription of the oxytrichid Tetmemena pustulata.... European J. Protistology. 49:272-282.
(2011) P. Madoni. Protozoa in wastewater treatment processess: A minireview. Italian. Journ. Zool. 78: 3-11.
(2007) D. H. Lynn The ciliated Protozoa. Springer Verlag.
(2007) W. Foissner, A. Chao, Laura A. Katz journal Biodiversity and Conservation, Volume 17, No 2, 345–363 - download PDF
(2004) N. G. Senler & I. Yildiz. Faunistic and Morphological Studies on ciliates from a Small Pond, with Responses of ciliate Populations to Changing Environmental Conditions - download PDF.
(2000) Illustrated guide to the Protozoa 2nd edition Society of Protozoologists. Allen Press, USA available on Amazon.com most recent Key to ciliates - check your library - excellent ref book.
(1999) W. Foissner, H. Berger and I. Schumberg. Identification and Ecology of Limnetic Plankton ciliates. Bavarian State Office for Water Management. 798 pages - download free ebook PDF
(1994) W. Foissner and S. Wölfl Revision of the genus Stentor Oken (Protozoa, Ciliophora) and description of S. araucanus from South American lakes. J. Plankton Res. 16: 225-289 - includes key to Stentors - PDF (see Dr. Foissner's other publications on his web site)
(1982) C. R. Curds British and Other Freshwater Ciliated Protozoa. Cambridge University Press.
(1972) H. Bick. Ciliated Protozoa World Health Organization, Geneva - free Ebook PDF
(1959) W.T. Edmondson Fresh Water Biology 2nd Ed. John Wiley, NY (Ward Whipple) - Keys to freshwater invertebrates (out of date but excellent illustrations). 1141 pages - Download PDF
(1953) W.J. Garnett Freshwater Microscopy. Constable & Co. Ltd London - Amazon.com $54.
Ciliates general reference - Wikipedia
Tear of a Swan - Lacrymaria olor - www.microscopy-uk.org
Paramecium (1985) ed H. D. Gortz Springer Verlag, NY
The Biology of Stentor (1961) Vance Tartar, Pergamon Press, NY
Freshwater Invertebrates - Ecology and General Biology (2010) eds J.H. Thorp & D.C.Rogers, Academic Press,NY - Excellent reference book - can purchase book from Elsevier
Pling Factory - In German use Google translation - Loxodes rostrum pictures and others
Microscopy from the very beginning - free book produced by Zeiss PDF
Boreal Science\Ward Science - can purchase some pond organisms
Quality Microscopes in Calgary - Zeiss dealer sells stereomicroscopes
Macrophotography.net - Forum on macro and photomicrography
Interested in Buying a Microscope - Visit New York Microscope Company
www.microscopeinternational.com
To all visitors, please enjoy a 5% discount with your order using the code 5BLCNP
Acknowledgements
I would like to acknowledge the assistance of Bruce Taylor in regards to identifying some of the ciliates, suggesting corrections and providing me with numerous ciliate research papers. You can visit his web site It Came from the Pond for more information about ciliates. Also view Bruce's videos of ciliates.
Note to teachers you have permission to use my pictures for teaching, I appreciate credit and\or link back to my web site if possible. For commercial use of my photos please contact me.
Authors Biography & Contact Information

Robert Berdan is a professional nature photographer living in Calgary, AB specializing in nature, wildlife and science photography. Robert retired from Cell\Neurobiology research to take up photography full time years ago. Robert offers photo guiding and private instruction in all aspects of nature photography and Adobe Photoshop training - including photomicrography, macrophotography.
Related Microscopy Articles by Robert Berdan on this web site
1. Photographing Stentors - A Large Unicellular Protozoan (ciliate) living in Freshwater
2. How to Collect and Photograph Water Bears (Tardigrades).
3. Tips on How to take Better pictures with a Microscope
4. Microscopic Pond Organisms from Silver Springs Calgary
5. Microscopic Life in Ponds and Rainwater - Pond Scum I
6. Photographing Microscopic Plant and Animal Life - Pond Scum II
7. Photomicrography and Video of Protozoa, Volvox and Rotifers
8. Home Microscopy Laboratory for Photomicrography
9. The Art & Science of Photomicrography with Polarized Light
10. Photographing Through a Microscope Photomicrography - Inner Space
11. Focus Stacking comparing Photoshop, Helicon Focus and Zerene
12. Rheinberg Filters for Photomicrography
13. Scanning Electron Microscopy - Photography
14. Photomicrographs of Diatoms from 1877 by John T. Redmayne
Email at: rberdan@scienceandart.org
Web site: www.canadiannaturephotographer.com
Phone: MST 9am -7 pm (403) 247-2457.
Click on the buttons below and share this site with your friends