
Photographing Rotifers
by Dr. Robert Berdan
July 24, 2018

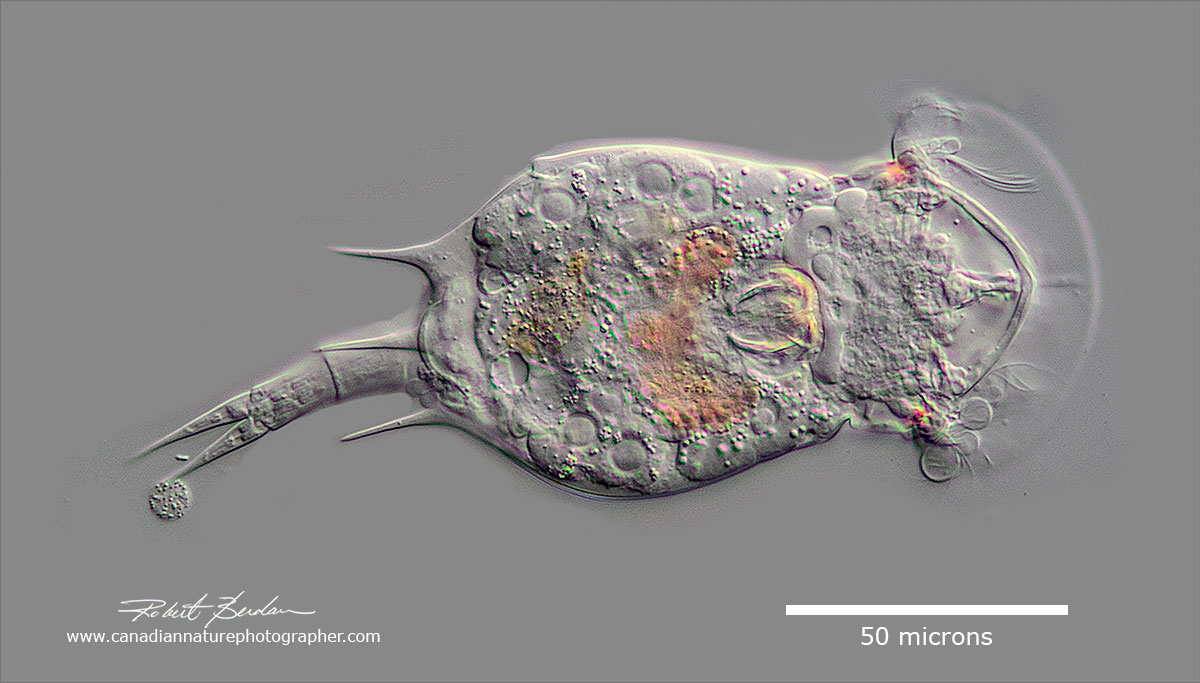
Euchlanis dilatata (Monogononta, Ploima, Euchlanidae) - a rotifer common in pond water (one micron = 1\1000 of millimetre) Differential Interference Microscopy (DIC) using a Zeiss Axioscope microscope.
Introduction to the Rotifers
An interest in Rotifers started after microscopist Anthony van Leeuwenhoek (1632-1723) described them in a letter from 1687. These microscopic organisms live in water including ponds, creeks, rivers, lakes, soil, water associated with moss, liverworts and lichens. They are also found in a more limited extent in saline waters. They range in size from 40 microns (0.04 mm) to about 2,000 microns (2 mm) in length so a light microscope is required to observe them. Rotifers have exisited for at least 35-40 million years as they have been identified in fossilized amber (Waggoner and Poinar, 1993). They only live about 1-2 weeks. These organisms are not well known though I think they should be.
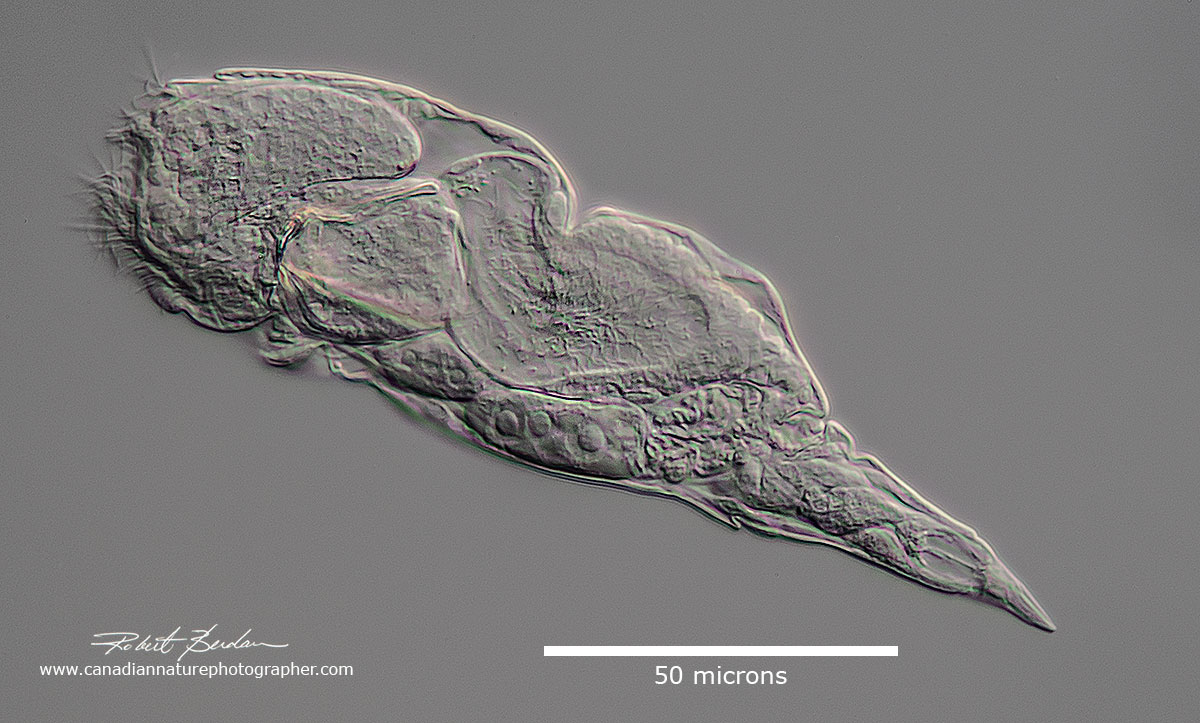
Squatinella rostrum (Rotifera: Monogononta, Ploima, Lepadellidae) note 2 red eyes on the side of the head. DIC microscopy.
It's estimated there are about 2000 species of rotifers, most live in freshwater. They can occur in numbers of up to 5000 per litre. They play a role in nutrient recycling, they are used in testing water toxicity, and are also used to feed fish larva and are even sold in some pet stores. In research they are used to study aging, suspended animation (anhydrobiosis), and are used as bioindicators of toxicity. They have been referred to as "Time travellers", because when in the dessicated state as a cyst or egg they can survive for decades without any signs of aging. Some have survived frozen in glaciers for over 24 thousand years (L. Shmakova et 2021 Current Biology).
Bedelloid rotifer photographed with positive phase contrast Olympus E- Microscope 200X

Bdelloid rotifer Negative Phase contrast - Nikon Optiphot microscope 200X

Bdelloid rotifer by Polarization Microscopy with a full wave plate - the muscles show up pink in colour 400X. Nikon Optiphot microscope.
Because few people have heard of rotifers I would like to share some pictures and information about these fascinating organisms. They live in my backyard, roof gutters, bird bath, tree lichen, moss and soil - in fact anywhere there is a temporary pool of water will usually be home to rotifers. They come in a wide variety of shapes and resemble aliens. Some groups of rotifer (Bdelloidea) have no males - only females yet they are able to thrive and evolve in the absence of sex.
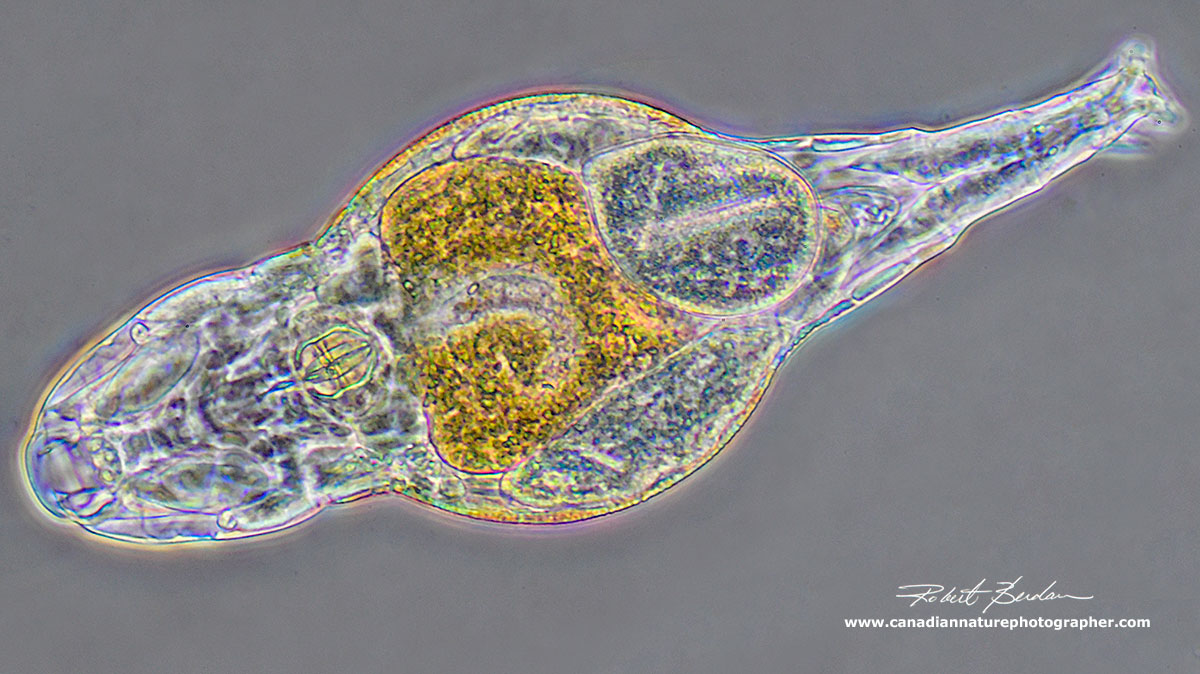
Side view of a rotifer with DIC microscopy showing a 3D like relief - the head is at the top left (Cyrotonia tuba).
There are three main groups of rotifers: 1) Bdelloidea - they can survive dessication and appear to consist of only females and reproduce asexually via parthenogenesis (unfertilized eggs). 2) Monogononta - they cycle between parthenogenesis and eggs that are fertilized and 3) Seisonidae a family of rotifers found only on the gills of a marine crustacean. I only show examples of rotifers from the first two groups. Below is a diagram of a model rotifer.

Above basic diagram of a female rotifer with the various internal organs labelled. Some rotifers lack some of these parts e.g. toes and show variations in morphology.
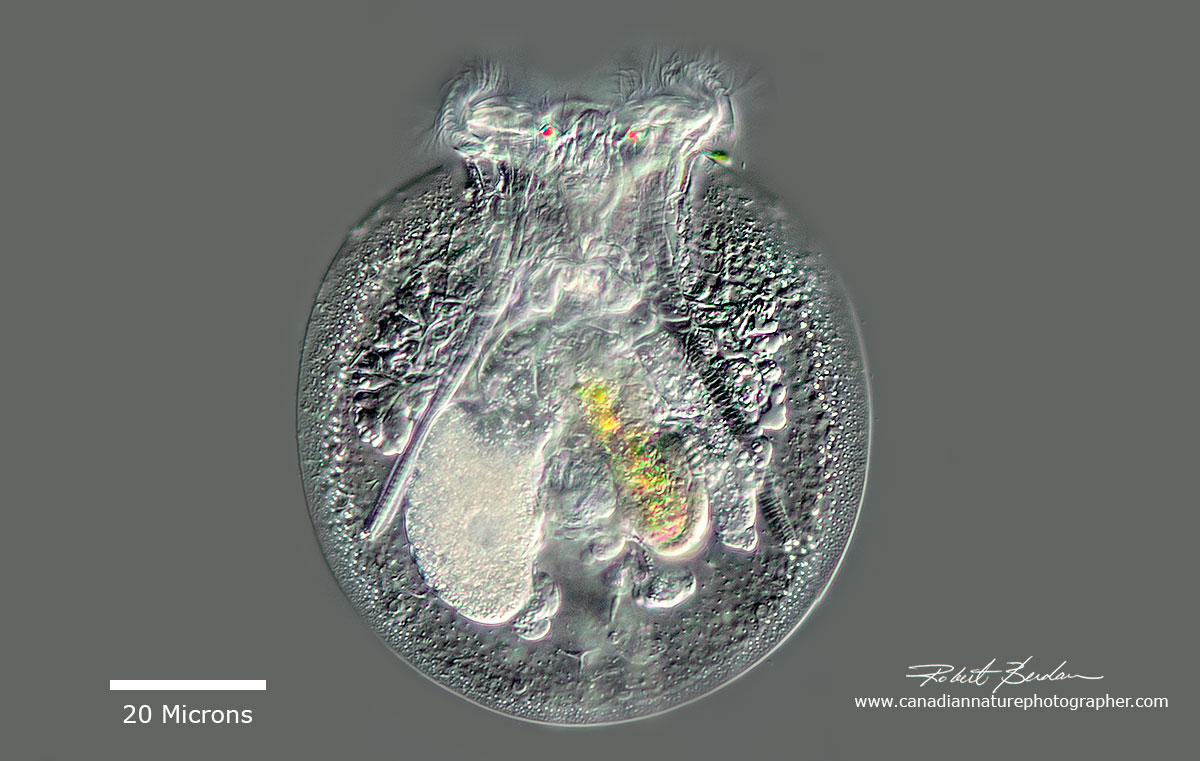
Testudinella patina - nicknamed "Turtle Rotifer" - has a telescoping foot that has contracted into the body, also note the two red eyes at the top. (Monogononta, Gnesiotrocha, Flosculariaceae).
Rotifers have other unusual characteristics. They possess a constant number of cells (900-1000) referred to as eutely. Their cells do not divide after they are born, growth occurs by changes in the size of the cells. Furthermore, most of their tissues are syncytial - that is many of the tissues share nuclei. Some tissue like the nerves and gut epithelium do have individual cells. Their body cavity is called a pseudoceol as it is not lined by an epithelium and functions as a hydrostatic skeleton, respiratory organ and circulatory system.
Finding and Collecting Rotifers

I collect rotifers and other protists from ponds near my home in Calgary. I use jars attached to a golf ball retriever to reach out into the water and a turkey baster from Walmart to collect sediment from around the edges of the pond as described in books by Tony Pattinson (The Freshwater Microscopist). A pair of rubber boots for wading is helpful.
I try to include some water plants in each jar as that is where I find most of the rotifers. A magnifying lens or stereomicroscope can be helpful to find the rotifers before placing them onto a microscope slide. Sometimes I use a centrifuge to concentrate specimens in a sample of water. When mounting the rotifers one has to be careful not to crush them when placing the overlying coverslip on a microscope slide. Sometimes I will add 4 small drops of vacuum grease (or vaseline) with a syringe on the corners of the coverslip and it press down gently over the sample. The grease helps prevent the coverslip from completely drying down and crushing the rotifers.

Large rotifer by Darkfield microscopy found in a local fish pond. Notommata copeus Ehrenberg 1834.
At the anterior end of many rotifers is a corona consisting a cilia which appear to rotate like a wheel resulting in their early name wheel animalcule (Rotifera latin rota - wheel). The cilia appear to rotate and function in locomotion and to draw food (bacteria, small algae, and yeast) into their mouth. The corona can be withdrawn into the animals lorica or shell and some of the rotifers can also move like an inch worm. See my video at the end of my article on Protozoa which shows rotifers crawling like an inch worm.
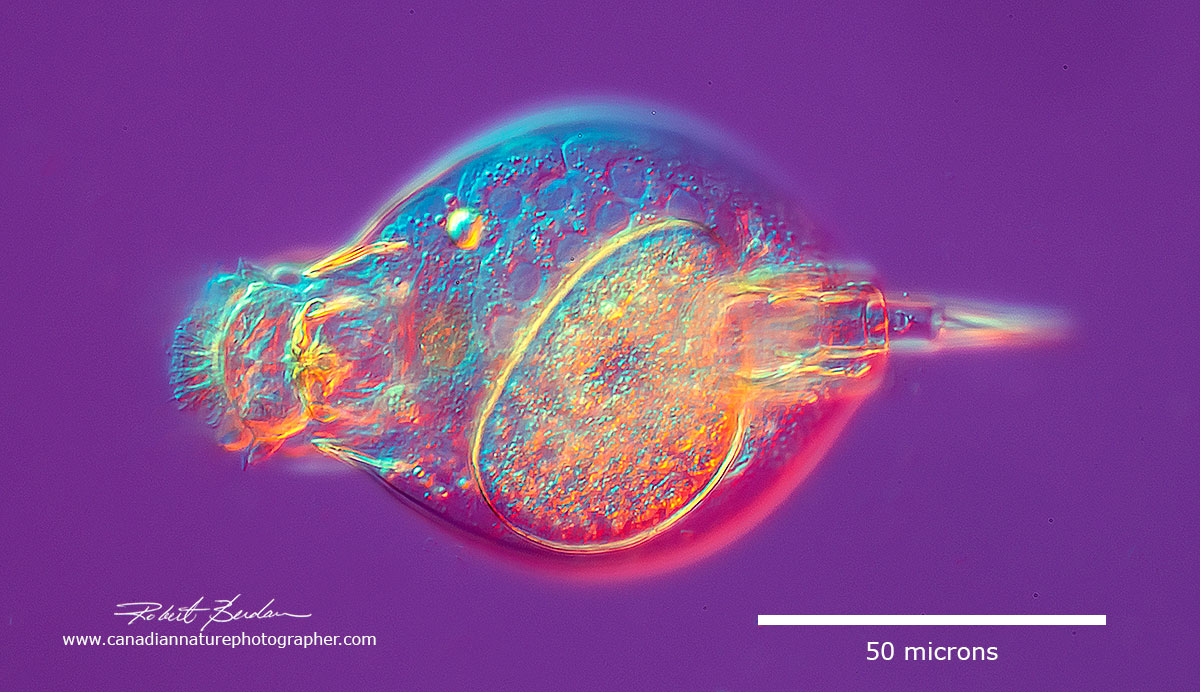
Lepadella ovalis with egg inside DIC microscopy. Monogononta, Plioma, Lepadellidae.

Lepadella ovalis - perfectly streamlined - DIC microscopy
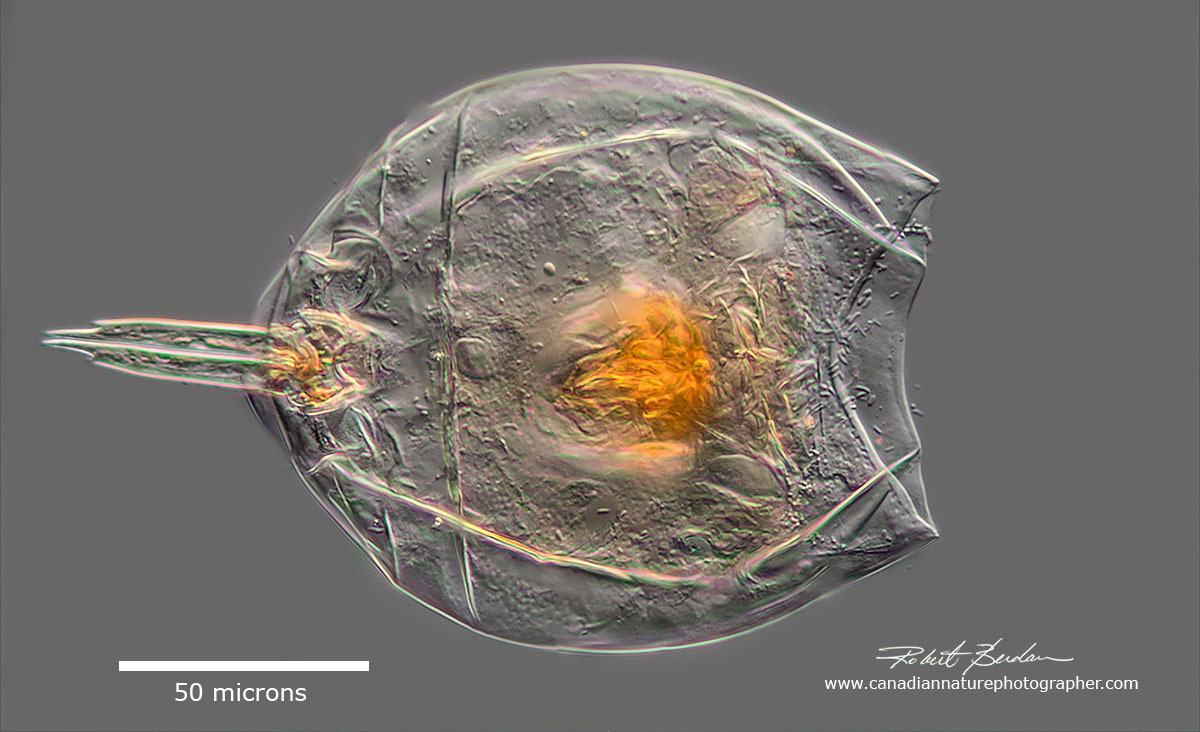
Lecan sp note the double foot and toes - head contracted into the lorica (shell). Monogononta, Lecanidae.
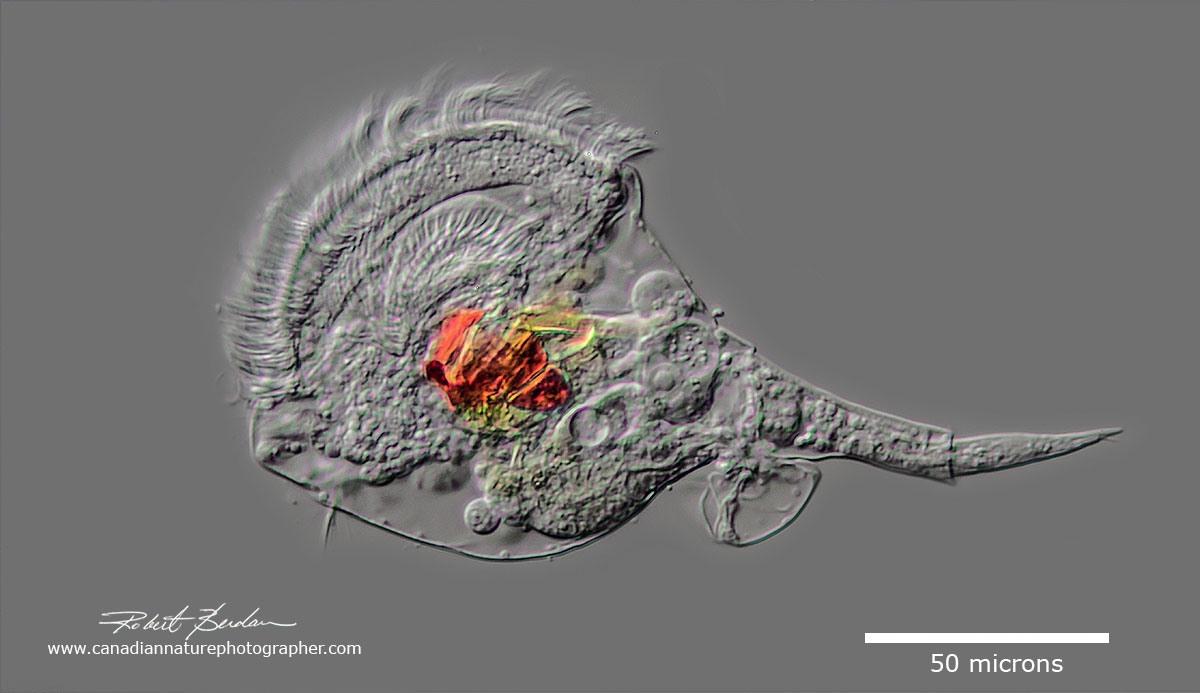
Unidentified rotifer species (possibly Microcodon clavis) with a single toe and wide ciliated corona.

Monommata arndti has a long foot Monogononta, Ploima, Notommatidae 200X
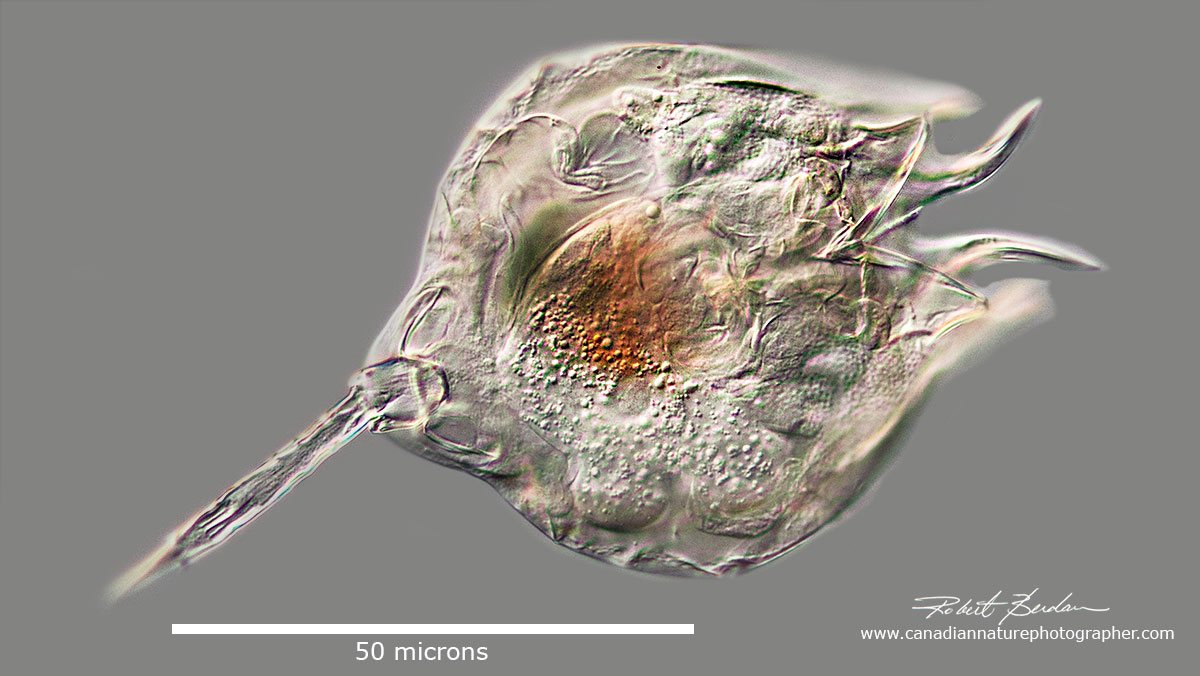
Lecane quadridentata (I think it was previously described as Monostyla quadridentata). It is nicknamed the "devil rotifer" because of the horn like projections from the lorica.

Bdelloid Rotifers - one with the corona beating and the other one with the corona contracted. 200X

Large Bdelloid rotifer found in moss - it appears to have an egg inside - corona retracted.

Euchlanis sp DIC microscopy - single red eyespot just barely visible

Euchlanis sp viewed by Dark field microscopy

Euchlanis sp viewed by DIC (differential interference contrast) microscopy

Euchlanis sp - have a two toes

Euchlanis sp with head retracted into its lorica

Rotaria neptunia - the most elongate rotifer known has 3 terminal toes and can extend its foot which "telescopes" increasing its body length almost 3X. Bdelloidea, Philodinidae, Rotoria.

Rotaria neptunia - a young rotifer can be seen inside the mother and can be identified by the extra trophi and red eyes.

Rotaria neptunia - showing 2 eyes and detail of the trophi which has 4 large teeth. The head and foot are often retractable into the trunk.

Bdelloid rotifer - note the red eyes

Bdelloid rotifer Philodina roseola. (Bdelloidea, Philodinida, Philodinidae)
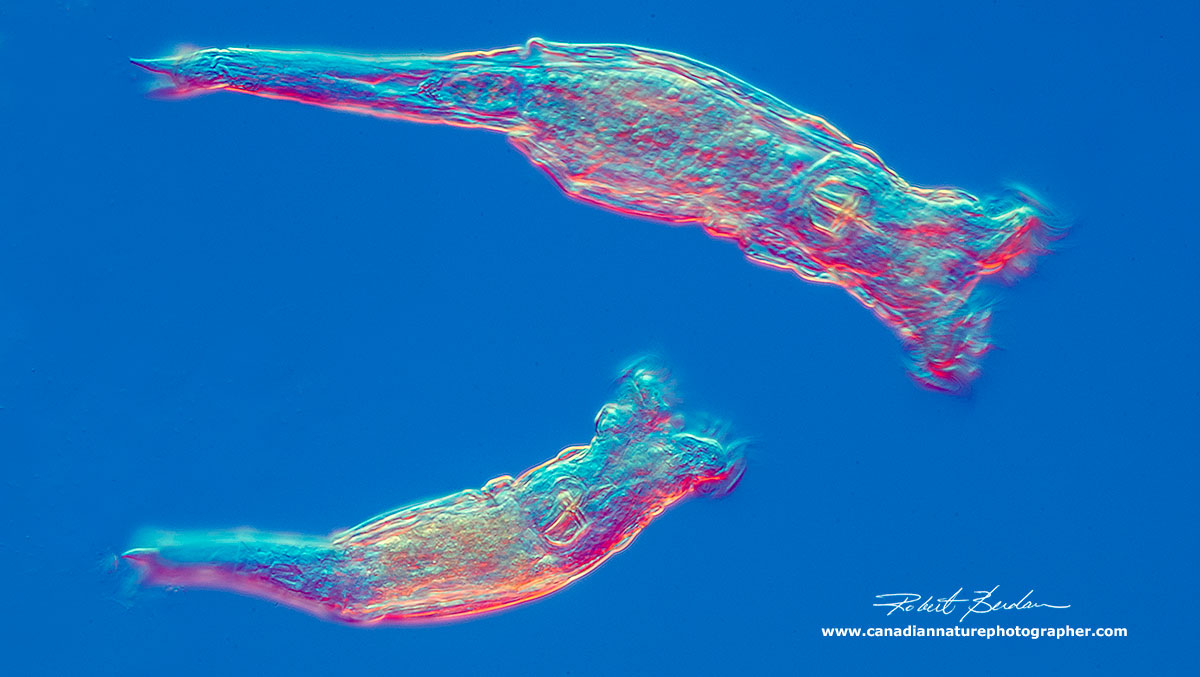
Philodina roseola swimming - note the two spurs at the posterior end - it is one of the most common species of rotifers. Bdelloidea, Philodinidae. The corona of cilia can provide locomotion or the rotifer can move by creeping like an inch worm. The head (rostrum) and the toes have special adhesive glands that can be used to anchor to the substratum so they can push or pull themselves along.
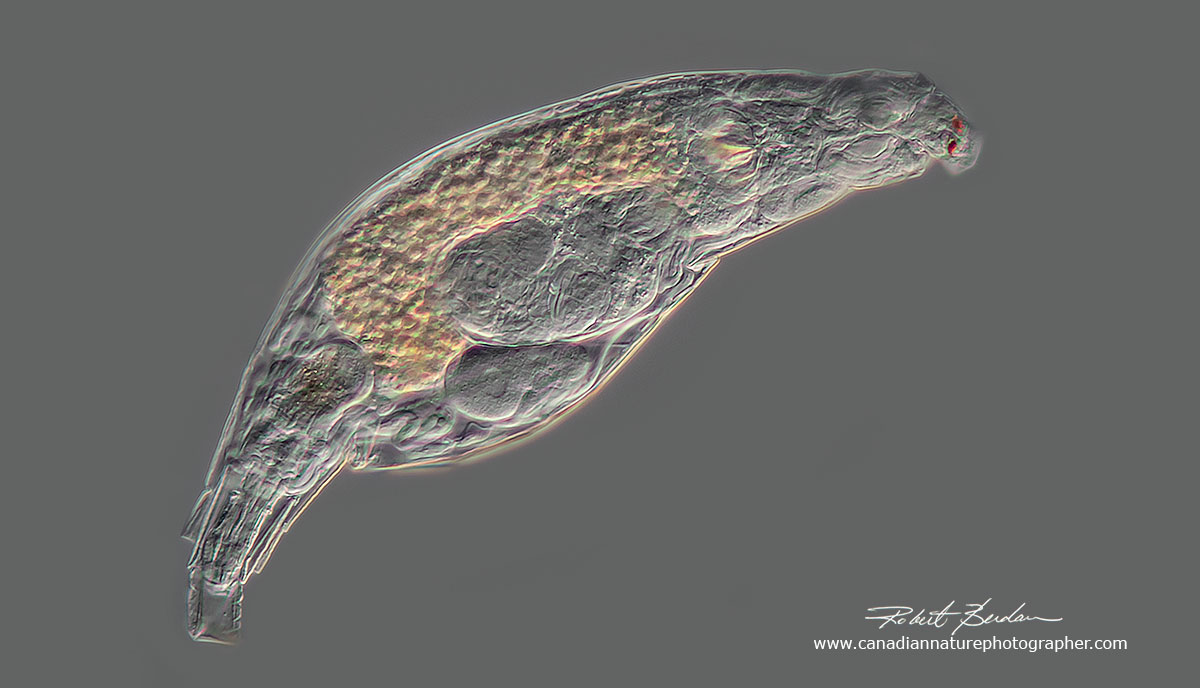
Large bdelloid rotifer with corona withdrawn and the rostrum containing the eyes extended
Trophi - are the hard chitinous parts of the masticatory (chewing) apparatus. It is often in motion when observing rotifers and can even be recognized in cysts and dead animals. The shape and organization of the trophi is used to taxonomically. It is situated in the ventral side of the pharynx (tube that extends from the mouth to esophagus) within the mastax which is composed of muscles that move the "jaws".

Bedlloid rotifer stretched out with corona retracted - note the trophi inside - DIC microscopy.
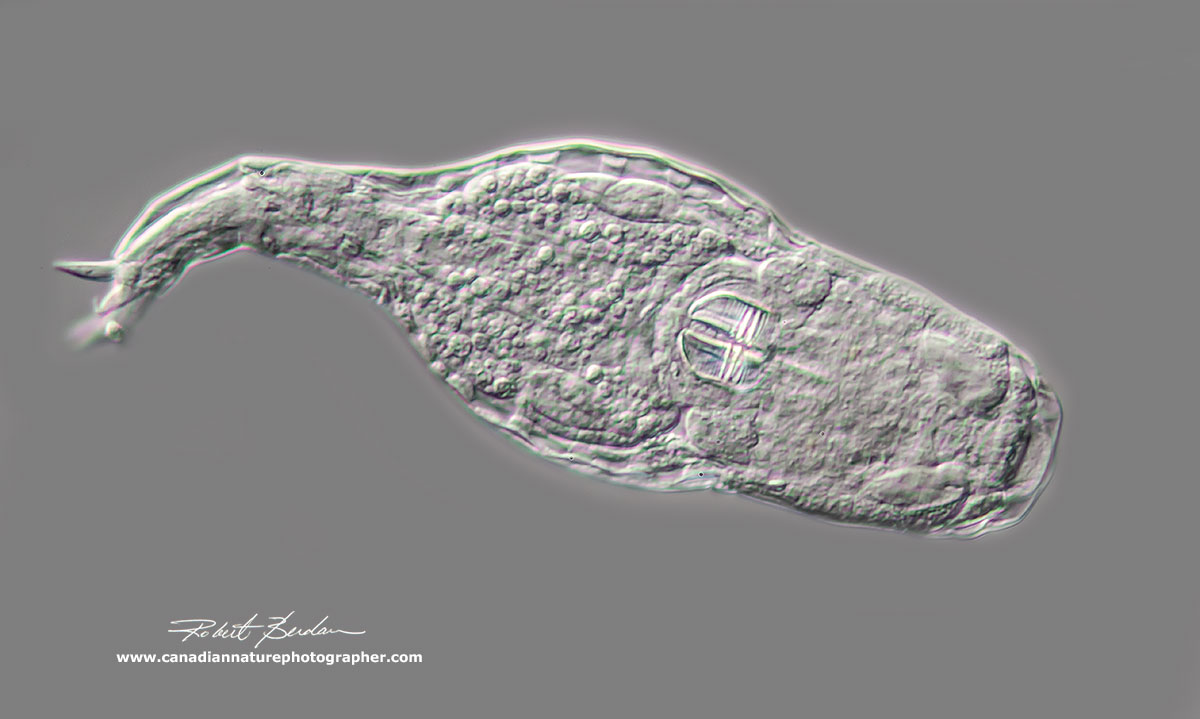
Bdelloid rotifer - the trophi and its teeth can be clearly seen inside the organism. This rotifer was crawling along like an inch worm. DIC microscopy.

Closeup of a Trophi from a bdelloid rotifer showing the chitinous teeth and other parts. Trophi play a role in grabbing prey and chewing food. In live rotifers they can often be seen moving inside the organism. The parts of the trophi are shown above - though a fulcrum is missing in Bedelloid rotifers. The jaws operate like forceps. Trophi attain their size and shape in the fully grown embryo and remain constant during the life of the rotifer. Rotifer biologists can isolate the Trophi by treating the organisms with Chlorox bleach which digests the rest of the organism. There are nine main types of trophi based on their shape and size of the elements. The shape of the trophi is important in rotifer taxonomy. For a detailed description of the various types of trophi see D. Fontaneto and W.H. De Smet (2015).

Euchlanis dilatata species? Side view (Eurotatoria, Plioma, Euchlanidae)
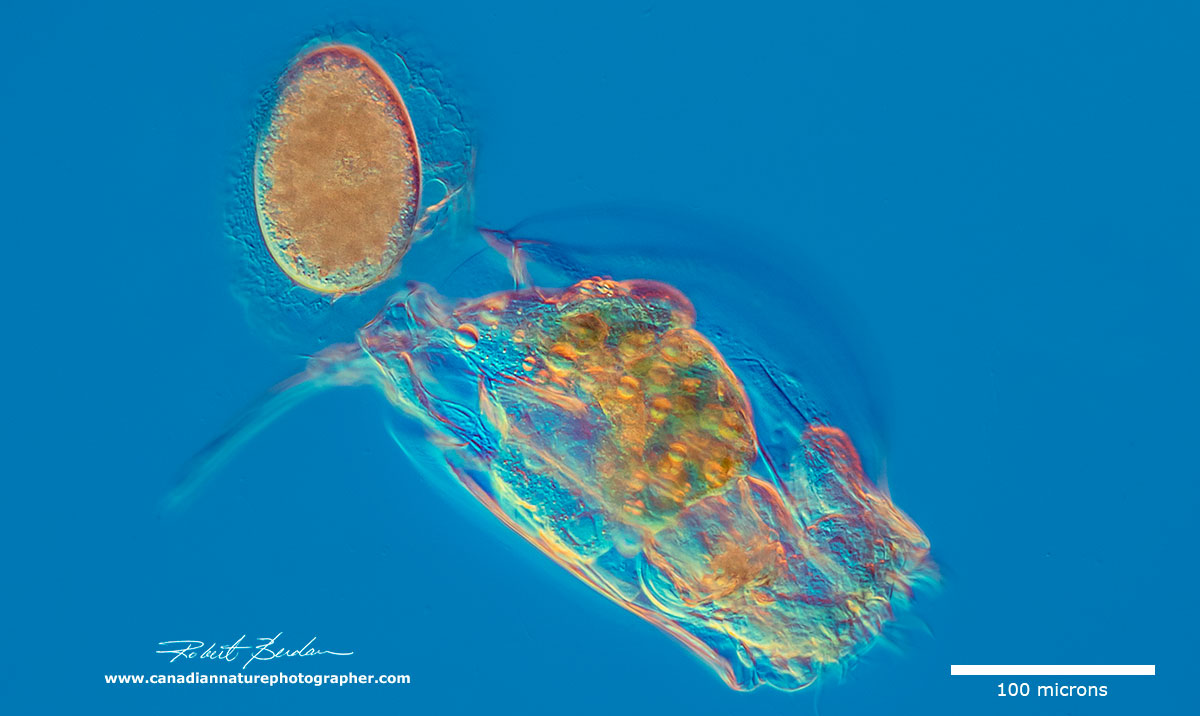
Rotifer releasing an egg (Euchlanis sp?)

Trichocerca sp? This is a small rotifer that is common in pond water.
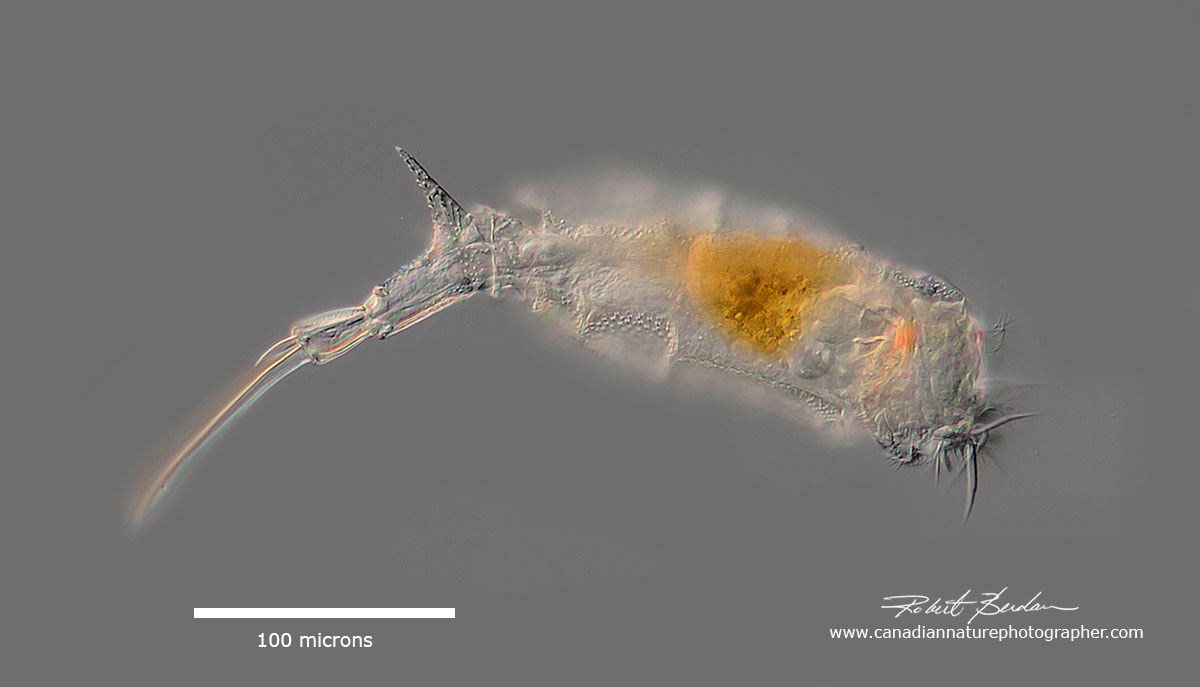
Trichotria sp (Trichotria tetractis? )- note the spur that looks like a shark fin. Eurotatoria, Plioma, Trichotriidae.
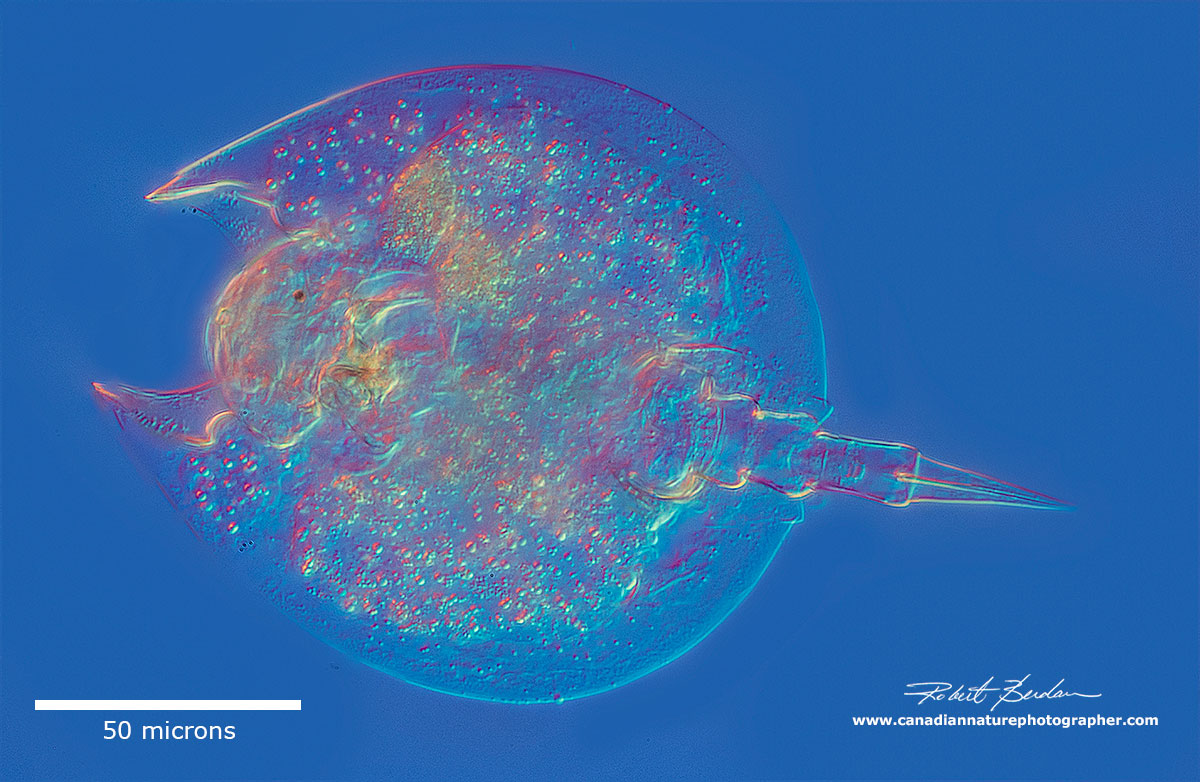 Lepadella ovalis - with head withdrawn into the lorica, photo stack.
Lepadella ovalis - with head withdrawn into the lorica, photo stack.

Large Bdelloid rotifer with young inside - note two trophi inside the animal.
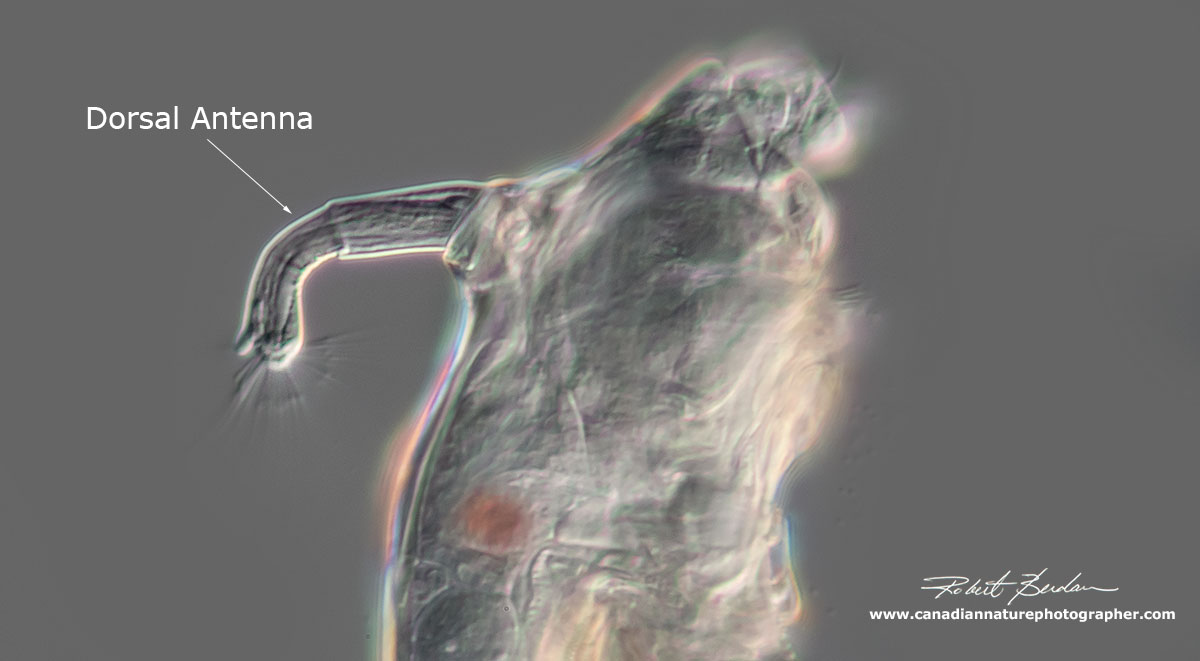
Note the Dorsal antenna which protrudes and retracts like a periscope - it's believed to be sensory in function.
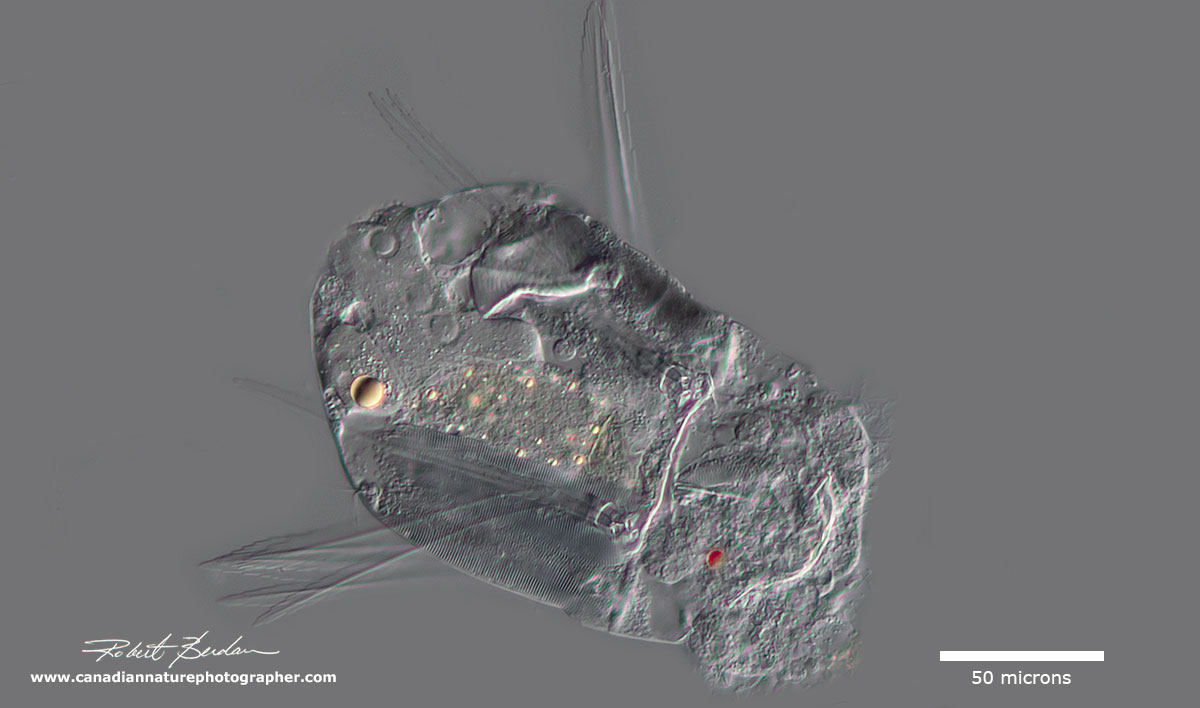
This Monogonota rotifer (Polyarthra remata?) has feather-like fins - the animal is partly compressed and unfortunately I damaged it when I mounted it.
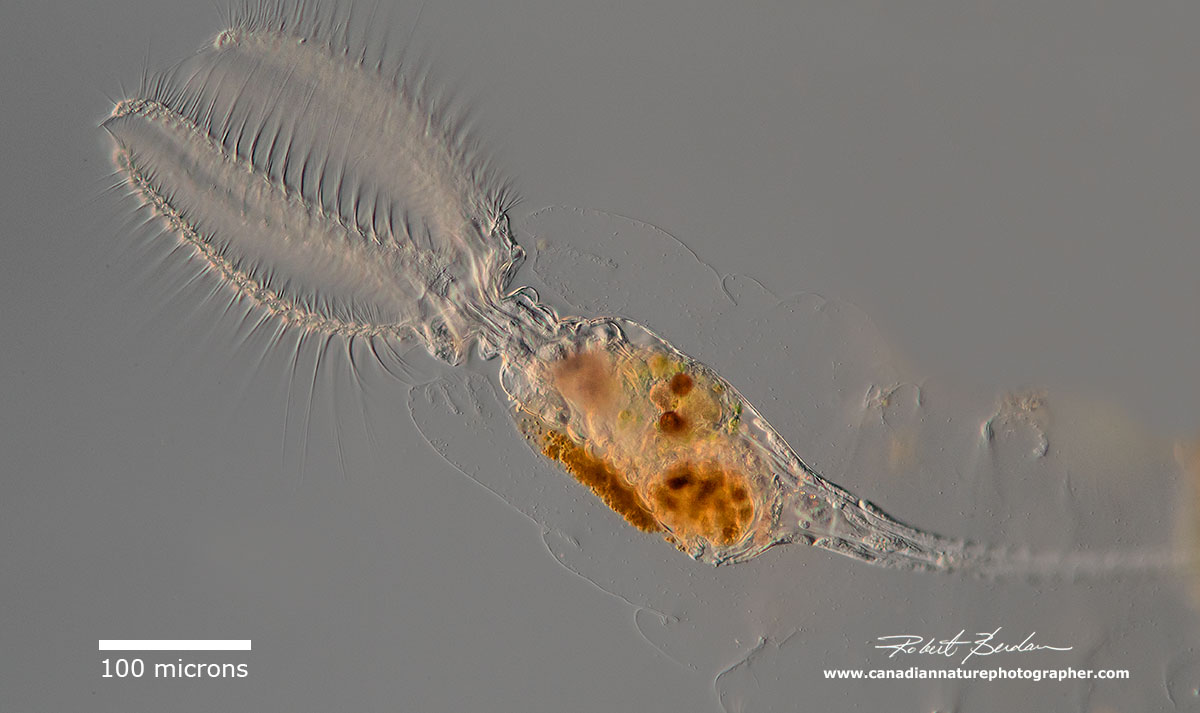
This sessile rotifer Stephanoceros fimbriatus (Monogonota, Gnesiotrocha, Collothecidae) was collected in a fish pond and found by itself attached to a branch of Bladderwort (Urtiularia). It was attached to the plant via a flexible retractable stalk. I was able to find it on an algae branch by viewing the algae with a stereoscope and then cutting the branch and putting it on a microscope slide.
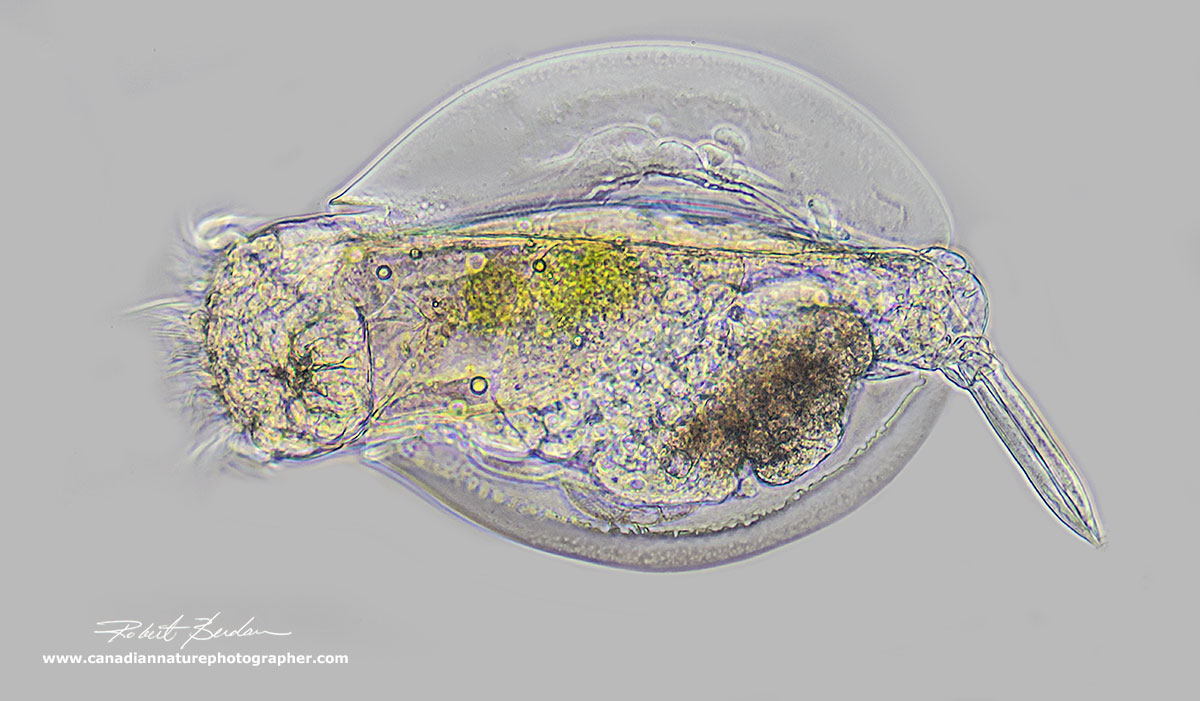
Rotifer by Phase contrast microscopy (Lapadella ovalis).
Brachionus Rotifers - may be available at your local aquarium or pet store

Brachionus is a genus of planktonic rotifers occurring in freshwater, alkaline and brackish water. I purchased a bottle of Brachionus manjavacas rotifers from a Pet store in Calgary ($30\bottle - Pisces Exotica Pet Emporium) - they are used to feed fish larva. I found two types of rotifers in this bottle; the one shown below is Brachionus. The lorica (shell) is pear-like shape. Brachionus is a Monogonont rotifer (Rotifera, Monogononta, Euroatoria, Poima, Brachionus sp). I placed a sample of the water in a shallow dish and viewed them under my stereoscope first and then selected them with an eyedropper to photograph with my light microscope. Brachionus sp are also used in toxicology, stress and aging research. Brachionus species can normally reproduce sexually and asexually (cyclical parthenogenesis). This Calgary pet store sourced the rotifers from Sustainable aquatics - for more information see their web site. They recommend storing the Rotifers in the fridge.
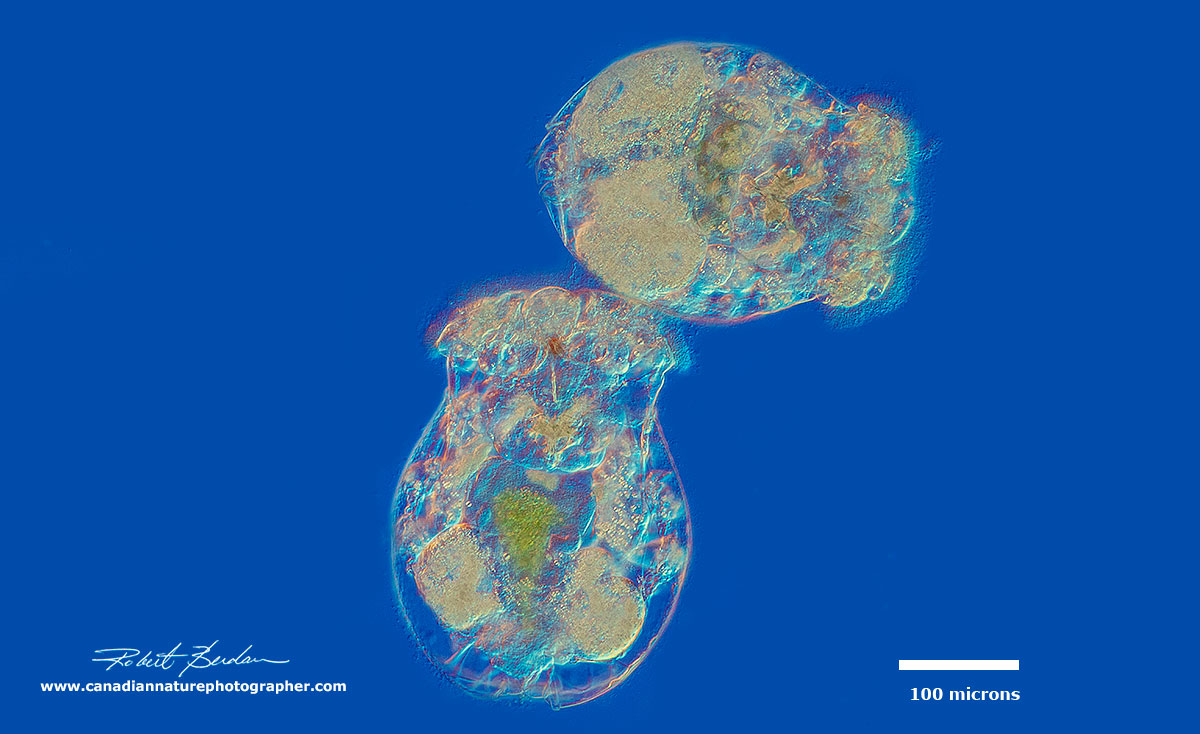
Two Brachionus manjavacas rotifers at low magnification - DIC microscopy

Brachionus manjavacas photographed using a combination of Rheinberg lighting and DIC microscopy. Note the red eye at the right.

Brachionus manjavacas head showing ciliated corona, a bit of the red eye spot - photo stack of 5 images.
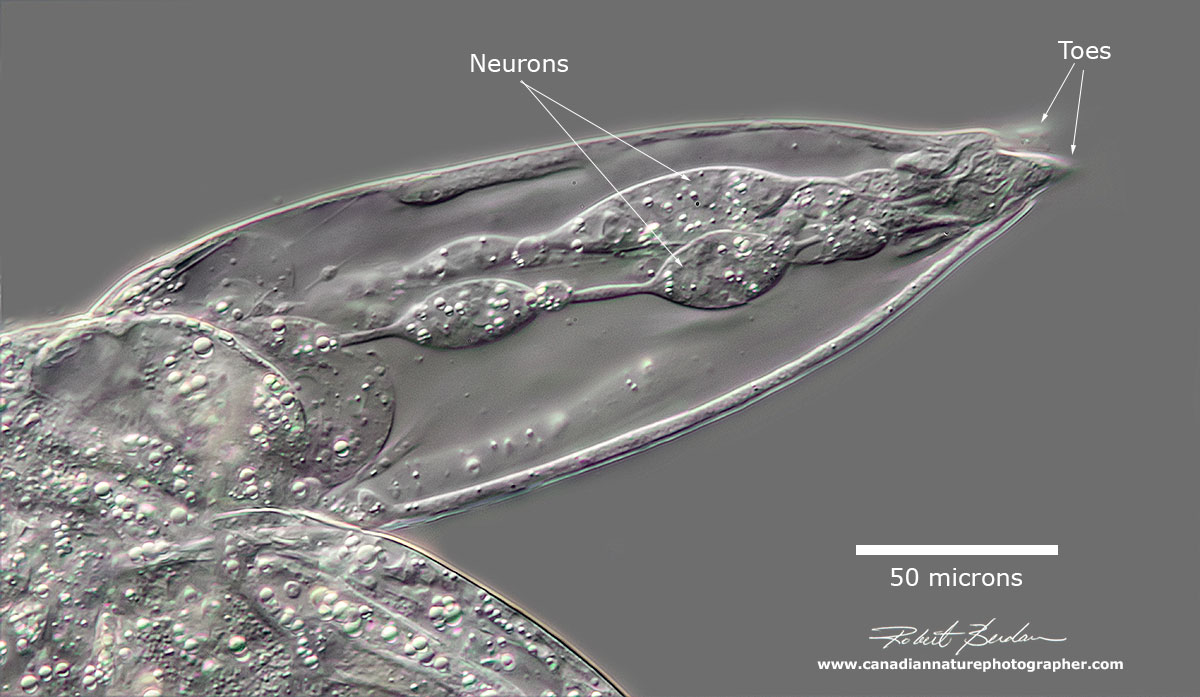
Brachionus manjavacas closeup of the foot showing cells (neurons or muscle) attached to the toes. DIC microscopy.

Brachionus manjavacas by Darkfield microscopy note the red eye
Male Rotifers

Female rotifer Colurella sp (Leadellidae). Males are rare and much smaller than females and in some groups of rotifers (Bdelloidea) males have never been reported. Males usually only live for a few days and have only one purpose to fertilize eggs - they don't or can't feed. The factors that control the appearance of males in a population is not well understood.
Cysts and anhydrobiosis - surviving dessication
Rotifers viewed with my microscope seem to be able to retract into their shell as soon as water under the coverslip starts to dry up and conversely they come back out within minutes of adding freshwater. The sugar Trehalose has been suggested to play an important role in anhydrobiosis of Tardigrades but it is absent in the Bdelloidea rotifers. Other molecules like heat shock proteins, chaperones (proteins that assist in protein folding), antioxidants etc may be more important. Anhydrobiosis in rotifers requires more research and it may have implications for space travel in the future.

Rotifer Cyst with trophi visible near the center - DIC microscopy
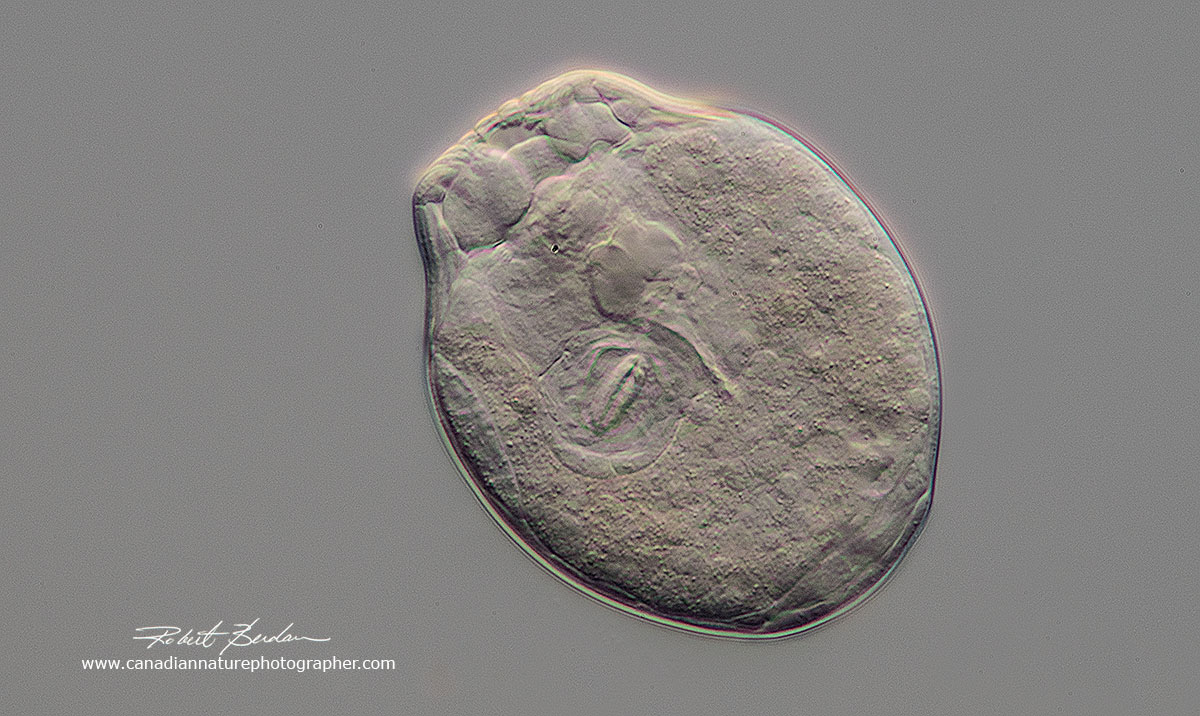
Rotifer cyst - you can see the Trophi inside the shell and in the photo below the rotifer coming out of the shell. Rotifers will retreat into their lorica or shell when I flatten the coverslip by removing water with a piece of filter paper that I touch to the edge of the coverslip. If I add water to the side of the coverslip - the rotifer appears to rehydrate within a few minutes and they come back out of their "shell".
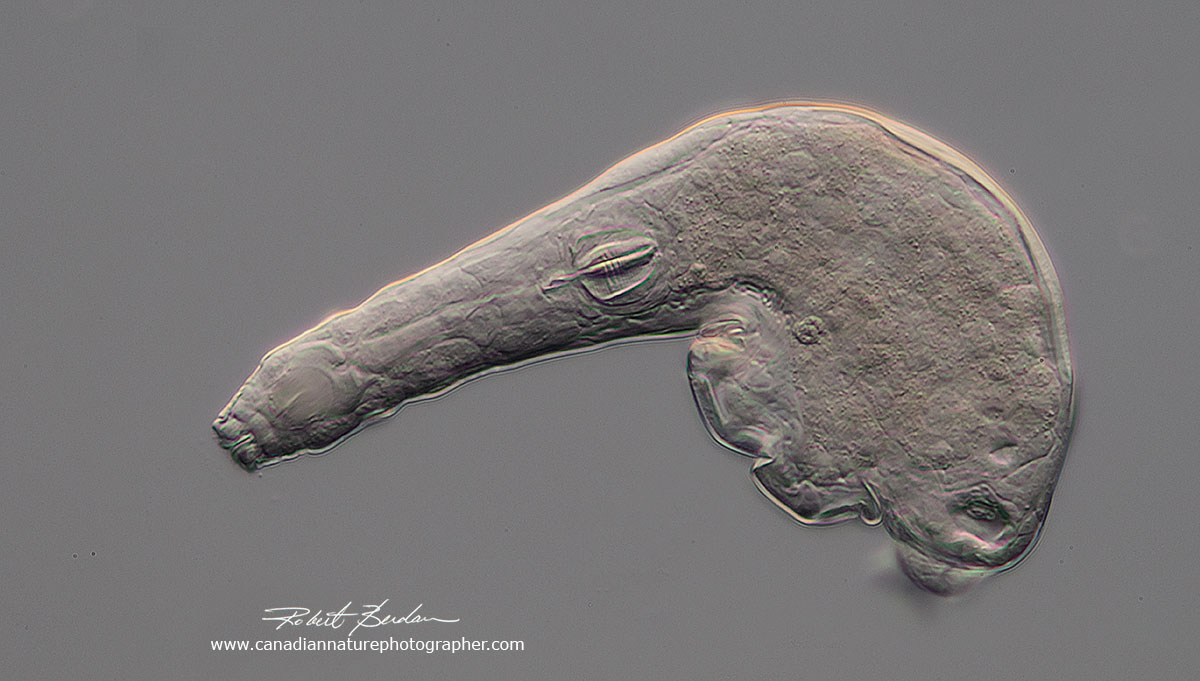
Rotifer coming out of the cyst within a few minutes of adding fresh pond water

Cyst belonging to a rotifer - the trophi is just visible through the shell. Rotifers can survive dessication in cyst form for long periods of time.
This is appears to be a rotifer tun - similar to the "tuns" formed by water bears and serves the same purpose - to survive dessication. Rotifers can survive for decades in this state and do not age or if they do - they age very slowly. One researcher likens these cysts or tuns to a "time machine". This picture resembles a tun from Macrotrachela quadricornifera Fig. 2 (Ricci and Fontaneto, 2009). Because Tardigrades also exhibit anhydrobiosis and form tuns it has been proposed that rotifer tuns be called a "xerosome" (Wallace et al. 2010) to distinguish them from those produced by Tardigrades.
Classification of Rotifers - Segers (2007)
Domain: Eukaryote (cells with a nuclear membrane)
Kingdom: Animalia
Clade: Platyzoa (monophyletic group in which members are thought to have evolved from a common organism).
Phylum: Rotifera
Class: Pararotatoira - single Family: Seisonidae - marine, obligatory sexual - species
Class: Eurotatoria
Subclass: Bdelloidea - Parthenogenic (develop from eggs without fertilization) - 461 species
Order: Adinetida
Order: Phildinavida
Order: Philodinida
Subclass: Monogononta - Intermittently sexual, but no males have been reported - 1570 species
Superorder: Pseudotrocha
Order: Ploima
Superorder: Gnesitrocha
Order: Flosculariacea
Order: Collothecacea
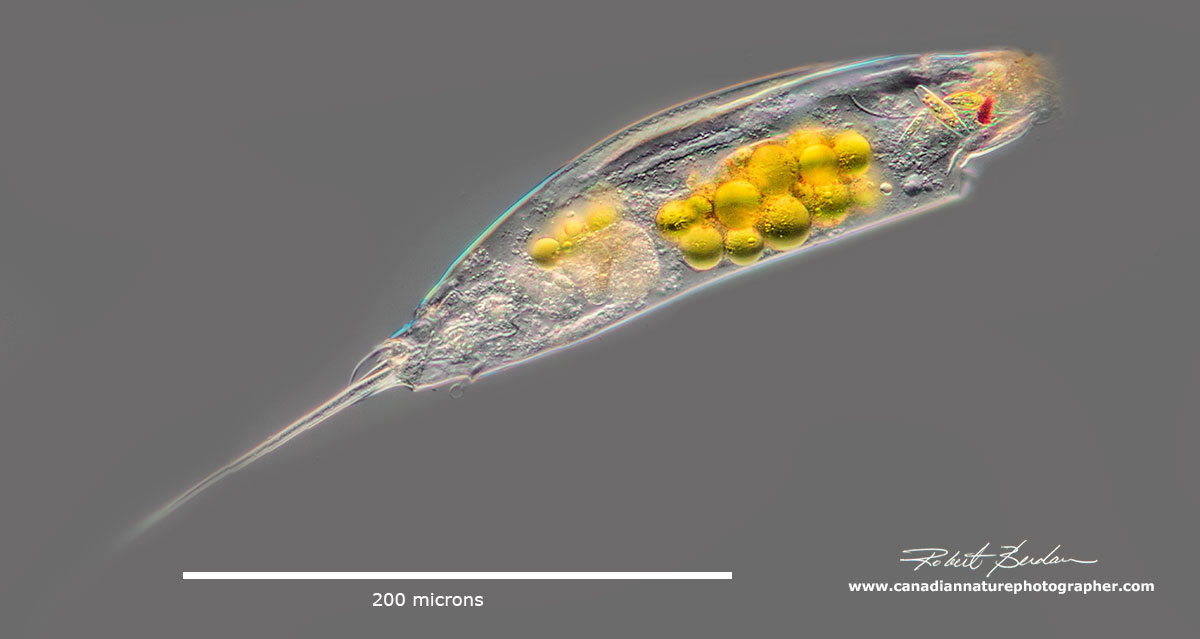
Trichocerca cylindrica focus stack by DIC microscopy

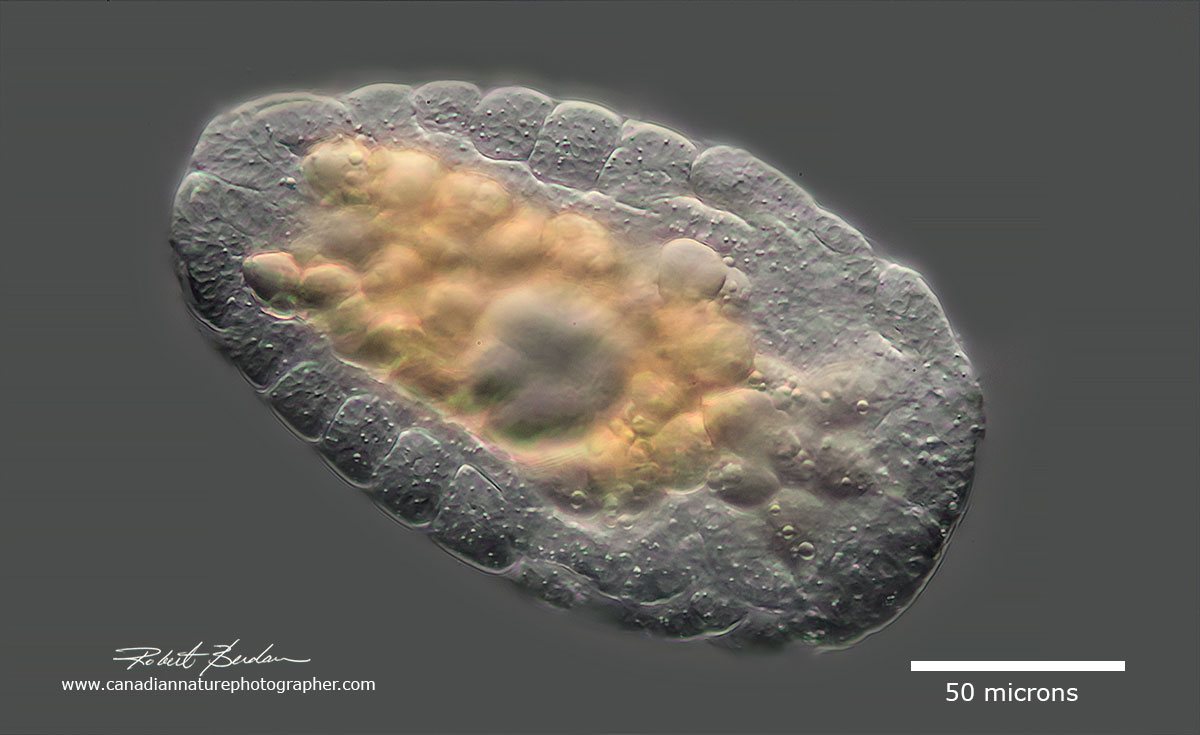
One of the largest rotifers Asplanchnopus multiceps with two chydorine chydoreds in the gut alonine and Chydorine chydorid (identity of Cladocerans courtsey of Dr. Russell Shiel University of Adelaide). Its transpacent carapace allows you to view the internal organs and cells with DIC. Epiphanes clavulata (Epiphanes: Epiphanidae: Ploima: Pseudotrocha: Monogononta: Eurotatoria)

Rotifer - Platyias quadricornis Ventral view of the Lorica - Stack of 15 images DIC microscopy (Monogonota, Pseudotrocha, Plioma, Bradionidae).
Photomicrography
All photographs in this article are of live rotifers collected from fresh water water near my home (except Brachionus sp purchased at a pet store). For details on how I took these pictures please see my article - Tips on How to Take Better Pictures with a Microscope - Photomicrography. I used a Nikon D500 and Nikon D800 cameras, Zeiss Axioscope with DIC, Bright field, and Darkfield lighting, and controlled my cameras with free software Digicam control. Images and video were captured onto an Alienware laptop and processed in Adobe Photoshop. The two biggest challenges is getting the rotifers to hold still and the shallow depth of field that occurs in microscopy at higher magnification. In many instances I stacked 2-12 images to increase the depth of field - for more information on this procedure see my article on focus stacking. The background colours can be changed arbitrarily using a full wave plate with DIC microscopy. Rotifers were isolated from plastic Petri dishes and collected with a glass pipette, plastic eppendorf pipette or a small Irwin loop (handle with a small wire loop) and placed onto microscope slides for photomicrography.

Testudinella patina - Turtle Rotifer DIC microscopy - a wave plate was used to change the background colour.
Summary
In this short introduction to Rotifers I tried to show some of their unusual shapes and features. They can be found in almost any body of freshwater and\or live in the film of water in soil, moss, lichens and bryophytes. The Bdelloids are interesting in that they are able to evolve in the absence of males and may acquire new genes through horizontal gene transfer (from DNA of surrounding bacteria, algae and other organisms). Their ability to survive dessication for decades like the Tardigrades (Water bears), Nematodes and other Protists living in pond water allows them to be found almost everywhere there is water. They play an important role in nutrient cycling and are used in research for toxicity testing and aging research. They are interesting to watch and photograph with a microscope and are found almost everywhere. If you don't own a microscope, no worries just come back and visit my web site as I will be posting more photomicrographs in the future but you can purchase a pretty good used microscope or even a new starter microscope for under $1000. RB
References (most can be found online)
(2017) A. Örstan and M. Plewka. An Introduction to Bdelloid Rotifers and their Study - www.quekett.org/starting/microscopic-life/bdelloid-rotifers
(2015) D. Fontaneto and W.H. De Smet - Chapter 4 Rotifera - In book: Handbook of Zoology, Gastrotricha, Cycloneuralia and Gnathifera. Volume 3, Publisher: de Gruyter Editors: Andreas Schmidt-Rhaesa pp 217- 300 - excellent recent review - download PDF
(2015) T. Pattinson. The Freshwater Microscopist Vol I and II. Blurb books.
(2012) E.F. Wilts, D. Wulfken, W.H. Ahlrichs and P.M. Arbizu. The musculature of Squatinella rostrum (Rotifera: Lepadellidae) as revealed by confocal laser scanning microscopy with additional new data onm its trophi and overall morphology. Acta Zoologica 93: 14-27.
(2010) R.L. Wallace, T. W. Snell and H.A. Smith. Phylum Rotifer Chapt 13 in Thorp and Covich's Freshwater Invertebrates. Academic Press, NW. 3rd edition. pp 225-271.
(2009) C. Ricci and D. Fontaneto. The importance of being a bdelloid: Ecological and evolutionary consequences of dormancy. Italian J. Zoology: 76: 240-249.
M. Plewka (2009) Lepadella ovalis - plingfacory.de
(2007) Segers. Annotated checklist of the rotifers (Phylum Rotifera) with notes on the nomenclature, taxonomy and distribution. Zootaxa 1564: 1-104, - download PDF.
(2006) R. L. Wallace. Rotifers: Exquisite Metazoans. Integ. and Comp. Biol. 42:660-667.
(1995) R.J. Shiel. A guide to identification of Rotifers, Cladocerans and Copepods from Australian inland Waters. Research Guide. Free PDF
(1974) A. Ruttner-Kolisko. Plankton Rotifers Biology and Taxonomy. Stuttgart, Germany.
(1959) W.T. Edmonson (ed) Fresh Water Biology 2nd edition, John Wiley. NY pg 420-507 - read online version free.
Online image based key for Rotifers - cfb.unh.edu/cfbkey/html/rotifers.htm
An Illustraded Key to the Planktonic Rotifers of the Laurentian Great Lakes - free PDF
Rotifer World Catalog - rotifera.hausdernatur.at
Plingfactory - see numerous photomicrographs of rotifers by M. Plewka (In german use Google translator in your browser)
Microscopy from the very beginning - free book produced by Zeiss PDF
Boreal Science\Ward Science - purchase rotifers for study by mail
Quality Microscopes in Calgary - Zeiss dealer sells stereomicroscopes
Macrophotography.net - Forum on macro and photomicrography
Acknowledgements
Note to teachers and students you have permission to use my pictures for teaching or school projects. I appreciate credit and\or link back to my web site if possible. For commercial use of my photos please contact me. Note species were identified using a variety of keys. Please regard species identifications with some reservation as I am not a Rotifer taxonomist just an Rotifer enthusiast. Thank you to Josh Grosse for pointing out some species errors I have made the corrections. I also thank Dr. R. Shiel (Invertebrate biologist) from Australia for pointing out some of my identification errors (rjsheil.com and https://www.adelaide.edu.au/directory/russell.shiel).
Authors Biography & Contact Information

Robert Berdan is a professional nature photographer living in Calgary, AB specializing in nature, wildlife and science photography. Robert retired from Cell\Neurobiology research to take up photography full time years ago. Robert offers photo guiding and private instruction in all aspects of nature photography and Adobe Photoshop training - including photomicrography, macrophotography.
Related Microscopy Articles by Robert Berdan on this web site
1. Photographing Ciliates
2. Photographing Stentors - A Large Unicellular Protozoan (ciliate) living in Freshwater
3. How to Collect and Photograph Water Bears (Tardigrades).
4. Tips on How to take Better pictures with a Microscope
5. Microscopic Pond Organisms from Silver Springs Calgary
6. Microscopic Life in Ponds and Rainwater - Pond Scum I
7. Photographing Microscopic Plant and Animal Life - Pond Scum II
8. Photomicrography and Video of Protozoa, Volvox and Rotifers
9. Home Microscopy Laboratory for Photomicrography
10. The Art & Science of Photomicrography with Polarized Light
11. Photographing Through a Microscope Photomicrography - Inner Space
12. Focus Stacking comparing Photoshop, Helicon Focus and Zerene
13. Rheinberg Filters for Photomicrography
14. Scanning Electron Microscopy - Photography
15. Photomicrographs of Diatoms from 1877 by John T. Redmayne
Email at: rberdan@scienceandart.org
Web site: www.canadiannaturephotographer.com
Phone: MST 9am -7 pm (403) 247-2457.